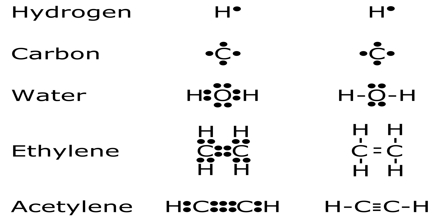
Major purpose of this lecture is to discuss on Chemical Bonding. A chemical bond results from strong electrostatic interactions between two atoms. The nature of the atoms determines the kind of bond. Each atom donates an electron resulting in a pair of electrons that are SHARED between the two atoms. For example, consider a hydrogen molecule, H2. When the two hydrogen, H, atoms are far apart from each other they do not feel any interaction.
Chemical Bonding