Mercury poisoning is a type of metal poisoning caused by mercury exposure. Symptoms vary according to the type, dose, method, and duration of exposure. Muscle weakness, poor coordination, numbness in the hands and feet, skin rashes, anxiety, memory problems, difficulty speaking, hearing, or seeing are some of the symptoms. Minamata disease is caused by high levels of methylmercury exposure.
When a person is exposed to high levels of mercury and the body is unable to effectively eliminate it, this is referred to as mercury poisoning. Mercury is a heavy metal that occurs naturally in three forms: elemental (or metallic) mercury, inorganic mercury compounds, and organic mercury compounds. Mercury in various forms can have varying toxic effects on the body.
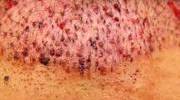
In children, methylmercury exposure can cause acrodynia (pink disease), in which the skin turns pink and peels. Long-term complications may include kidney problems and cognitive decline. The long-term effects of low-dose methylmercury exposure are unknown.
There are several ways in which mercury poisoning can occur:
- Occupational exposure: Certain industries, such as mining, refining, or the manufacture of mercury-containing products, may expose workers to high levels of mercury vapour or dust.
- Environmental exposure: People who live near industrial facilities or polluted areas may be exposed to mercury through air, soil, or water pollution. This can occur as a result of activities such as coal combustion or waste incineration, both of which emit mercury into the atmosphere.
- Consumption of contaminated food and water: High levels of methylmercury, an organic form of mercury, can be found in certain types of fish and shellfish. Consuming contaminated seafood can result in mercury poisoning, especially if done frequently or in large quantities.
Mercury exposure can take the form of metal, vapour, salt, or organic compound. The majority of exposure comes from eating fish, amalgam-based dental fillings, or occupational exposure. Higher up the food chain in fish, the more mercury there is, a process known as biomagnification.
Poisoning as a method of suicide is a less common occurrence. Human activities that release mercury into the environment include coal combustion and gold mining. Mercury tests for blood, urine, and hair are available, but they do not correlate well with the amount in the body.
The symptoms of mercury poisoning depend on the form of mercury involved and the level and duration of exposure. Some common symptoms include:
- Neurological problems: Memory loss, difficulty concentrating, irritability, tremors, coordination problems, and sensory disturbances such as numbness or tingling.
- Gastrointestinal issues: Nausea, vomiting, diarrhea, and abdominal pain.
- Kidney damage: Mercury can cause damage to the kidneys, leading to impaired kidney function and potentially kidney failure.
- Respiratory problems: In cases of inhalation of mercury vapor, symptoms may include coughing, chest tightness, and difficulty breathing.
- Cardiovascular effects: High levels of mercury can affect the heart and blood vessels, leading to increased blood pressure, heart rate irregularities, and potential cardiovascular damage.
Prevention includes eating a low-mercury diet, removing mercury from medical and other devices, disposing of mercury properly, and not mining any more mercury. Chelation with either dimercaptosuccinic acid (DMSA) or dimercaptopropane sulfonate (DMPS) appears to improve outcomes in those with acute inorganic mercury salt poisoning if given within a few hours of exposure. Chelation may be beneficial for those who have had long-term exposure. Mercury poisoning among children has been reported to be as high as 1.7 per 100 in certain fishing communities.
Prevention is key in avoiding mercury poisoning. Some preventive measures include:
- Minimizing exposure to mercury in the workplace by following safety guidelines and using appropriate protective equipment.
- Being aware of potential sources of mercury in the environment and taking precautions to limit exposure, such as avoiding contaminated water bodies or consuming fish with high mercury levels.
- Proper disposal of mercury-containing products, such as thermometers or fluorescent light bulbs, to prevent environmental contamination.