Executive Summary
During the recent years the world of business has changed vastly. We the people of different walks of life cannot be able to imagine that what is going to be the future picture if this change continues rapidly like this speed. Today there are very much competition in-between them and for that reason each and every business organization are trying to provide better service for their customers and trying to marketing their product to the customers better than the competitors.
We have learnt much about the Principles of Marketing .But don’t now how they are applied in the real lives. To find this out i.e. how they are applied in the real lives we were given this project.
For completing this project we needed information and to extract information we had to choose a mobile phone service company. For this we have chosen a company both having a good deal of customers and facing stiff competitions in their own business environments.
Taking information from them we have made our efforts to point out what are the marketing strategies and activities that these two companies sketch and undertake to remain unique, facing competitions and throwing challenges to the counterparts.
We tried our best to fetch all the possible information and include them in a relevant manner.
Marketing
“Marketing is the social and managerial process, by which individuals or groups obtain what they need and want through creative and exchanging products and values with others by considering social well being.”
Philip Kotler
Gray Armstrong
Principles of marketing
Marketing, the process by which a product or service originates and is then priced, promoted, and distributed to consumers. In large corporations the principal marketing functions precede the manufacture of a product. They involve market research and product development, design, and testing.
Marketing concentrates primarily on the buyers, or consumers. After determining the customers’ needs and desires, marketers develop strategies that are designed to educate customers about a product’s most important features, persuade them to buy it, and then to enhance their satisfaction with the purchase. Where marketing once stopped with the sale, today businesses believe that it is more profitable to sell to existing customers than to new ones. As a result, marketing now also involves finding ways to turn one-time purchasers into lifelong customers.
Marketing includes planning, organizing, directing, and controlling the decision-making regarding product lines, pricing, promotion, and servicing. In most of these areas marketing has overall authority; in others, as in product-line development, its function is primarily advisory. In addition, the marketing department of a business firm is responsible for the physical distribution of the products, determining the channels of distribution that will be used, and supervising the profitable flow of goods from the factory or warehouse.
Marketing Concept
The marketing concept holds that achieving organizational goals depends on knowing the needs and wants of target markets and delivering the desired satisfaction better than the competitors do. Example-
“Love the Customers and not the product”
The concept rests on four pillars-
- Target market.
- Customers need.
- Integrated marketing.
- Profitability.
Core marketing concepts are-
- Needs
Need means the basic human requirements or the state of feel deprivation. People need food, air, water, clothing and shelter to survive. People also have strong need for recreation, education .
- Wants
Needs become wants when they are directed to specific objects that might safety the need for example; an American needs food hamburger and they need rice to meet their hunger needs.
- Demands
Are wants that are backed by buying power? Many people want a Mercedes only a few are able and willing to buy the one
- Values
A combination of quality, service and price (QSP), called the customer value triad. A ratio between what the customer gets – benefits and what he gives – costs.
- Customers Satisfaction
Customers Satisfaction Depends on the product s perceived performance in delivering value relative to buyer s expectations.
- Quality
Quality is the totality of features and characteristics of a product or service that bears its ability to satisfy stated or implied needs
- Exchange
Exchange is the act of obtaining a desired object from someone by offering in return
- Transactions
Transactions are the trade of values between two parties
- Relationships
- Markets
Market means the set of all actual and potential buyers of a product or service. In view point of marketing market may be-
- Target Market
- Market Place
- Market Space
- Meta Market
- Marketers
Marketing Strategies
Marketing strategy is the marketing logic by which the business unit hopes to achieve its marketing objectives
Strategic planning
Strategic planning is the process of developing and maintaining a strategic fit between the organization’s goal and capabilities and its changing market environment. It involves the following-
- Defining a clear company mission.
- Setting supporting objectives.
- Designing a sound business portfolio.
- Coordinating functional strategies.
Most large companies consist of four organizational levels. They are: corporate level, divisional level, business unit level and product level.
Step: 01
Defining a market-oriented mission
Mission statement is a statement of the organization’s purpose- what it wants to accomplish in the larger environment. It acts as an invisible hand that guides the people in the organization to do their activities. Good mission have three major characteristics. They are as follows-
- There must be limited number of goals.
- It must stress on the major policies and values that the companies want to honor.
- It must define the major competitive scope.
Beside this management must consider some aspect before making the mission statement. These aspects are-
- Mission statement should not too broad or too narrow.
- Mission should be realistic.
- Mission should be specific.
- Mission should fit the market environment.
- Mission should be based on its distinctive capabilities.
- Mission statement should be motivating.
Step: 02
Setting company objectives and goals
The company’s mission statement should be turned into detailed supporting objectives for each level of management. Generally objectives are those goals, which are specific with respect to magnitude and time. These objectives must match with the mission statement. The objectives must be-
- Hierarchical.
- Quantitivly Expressed.
- Consistent.
- Realistic.
Step: 03
Designing the business portfolio
Business portfolios are the collection of business and products that make up the company. Business portfolio planning involves two steps.
- The company must analyze its current business portfolio and decide which business should receive more, less or no in investment.
- It must shape the future portfolio by developing strategies for growth.
Analyzing the current business portfolio
The major activity in strategic planning is business portfolio analysis. Portfolio analysis is a tool by which management identifies and evaluates the various businesses making up the company. By analyzing the current business portfolio management put their strong resources into their more profitable business and phase down or drops its weaker business.
In this case the first step is to identify the strategic business unit (SBU). SBU is a unit of the company that has separate mission and objectives and that can be planned independently from other company business. Each business unit must have some characteristics. They are as follows-
- Independent planning for each SBU.
- Each SBU must have own set of competitors.
- There must be an independent manager responsible for each SBU.
In this case the next step is to assess the attractiveness of their each business unit. There are in this case the next step is to assess the attractiveness. The best-known portfolio-planning method was developed by the Boston Consulting Group, a leading management consulting firm.
Growth share matrix approach
A portfolio planning method that evaluates a company’s strategic business units in terms of their market growth rate & relative market share. On the horizontal axis, relative market share serves as a measure of company strength in the market. The growth share matrix defines four types of SBUs
Stars: stars are high-growth, high share businesses or products. They need heavy investment to finance their rapid growth. The marketing strategy for this is to hold the market share to maintain the profit margin in the competitive market.
Star |
Question Mark |
Cash Cow |
Dog |
Cash cow: cash cow is low-growth, high-share businesses or products. They produce a lot of cash that the company can use to pay the bills and to support the other business. The marketing strategy for this is to harvest the products or business.
Question mark: They are low-share business until in high growth marketer need to identify the reason behind their low share and should try to build into stars.
Dogs: dogs are low-share low-growth businesses and products. In this case the management must decide whether to maintain the business or terminate the business.
Problems of matrix approaches
This approach has some limitations. They are as follows-
- They can be difficult, time consuming and costly to implement.
- This approach focuses on classifying current business but provide little advice for future planning.
Developing strategy for growth and downsizing
On the basis of the current business portfolio the marketer decide which business needs more investment and which need require less. Beyond evaluating the current businesses the marketer must identify some strategy regarding growth. There are four types of growth strategies available for the marketer. One useful device for identifying growth opportunities is the product / market expansion grid.
Product / market expansion grid
Product / market expansion grid is the tool for identifying growth opportunities through market penetration, market development, product development, & diversification.
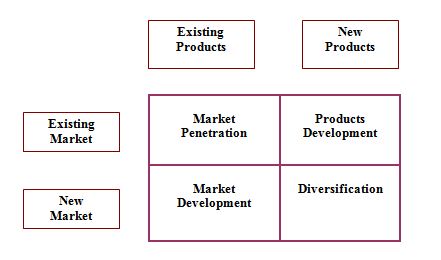
Figure : Product / Market Expansion Grid
Market Penetration
Market penetration is the strategy for company growth by increasing sales of current products to current market segments without changing the products. The different ways are-
- To induce the customer to buy more.
- Try to attract competitor customers.
- Try to identify the new ways of products
Market Development
Market development is the strategy for company growth by identifying & developing new market segments for current products. For example marketer could review their new demographic markets. The different ways are-
- Identify potential user group in the current market.
- Identify additional channel of existing products.
- Try to launch the products in new location.
Products Development
Products development is the strategy for company growth by offering modified or new products to current market segments. For example the marketer of fast food store offering launch box along with their fast food assortment. The marketer can develop new product by changing their features, quality etc.
Diversification
Diversification is the strategy of the company to start up a business, which are not similar to their current business products or markets. For example, we can say a textile company introduces a business regarding cosmetics. There are three types of diversification. They are as follows-
a) Concentric diversification
The company could seek new products that have technological or marketing synergies with existing product lines, ever though the new products themselves may appeal to a different group of customers.
b) Horizontal diversification
The company might search for new products that could appeal to current customers even though the new products are technologically unrelated to its current line.
c) Conglomerate diversification
The company might seek new business that has no relationship to its current technology, products or markets.
Companies must not only develop strategies for growing their business but also strategies for downsizing. The company must also carefully prune, harvest, divest of their old business in order to real ease needed resources and reduce costs.
Strategic planning and small businesses
Small businesses can also benefit from sound strategic planning. There are some steps needed to achieve the goals. They are as follows-
- Identify the major elements of business environment in which the organization has operated over the past few years.
- Describe the mission of the organization in terms of the nature and functions for the next two years.
- Explain the internal and internal forces that will impact the mission of the organization.
- Identify the basic driving forces that will direct the organizational features.
- Develop a set of long term objectives that will identify what the organization will become in the future.
- Outline is a general plan of action that defines the logistical financial and personal factors needed to integrate the long term objectives in to the total organization.
Role of marketing in strategic planning
Marketing plays a key role in the company’s strategic planning in several ways.
First, marketing provides guiding philosophy.
Second, marketing provides inputs to strategic planners by helping to identify attractive market opportunities and by assessing the firm’s potential to take advantage of them.
Third, marketing design strategies for reaching the unit’s objectives.
Relationship with customers
To succeed and to meet the intense competition need to develop the customer’s relationship. But before providing satisfaction needed to understand the needs and wants of the customers. And this process involving the following three steps-
- Market Segmentation
- Target Marketing
- Market Positioning
Market segmentation
Dividing a market into distinct group of buyers who have distinct needs, characteristics, or behavior and who might require separate products or marketing mixes. Segmentation can be possible on the basis of geographic, demographic, psychographics and behavioral factors. Every market has a segment but not all ways of similar way to a given set of marketing efforts.
Target marketing
The next step is to identify which market segments they want to enter. It is the process of evaluating market segments attractiveness and selecting one or more segments to enter. On the basis of the financial capabilities of the marketer they decide how many segments they enter. Most companies enter a new market by serving a single segment, and if this proves successful, they add segments.
Market positioning
The next step is to occupy a position in the mind of the customers. For each total segment, establish and communicate the key distinctive benefit of the offering. Basically it means that arranging for a product to occupy a clear, distinctive and desirable place relative to competing products in the minds of target consumers. Marketers want to develop unique market position for their products. If a product is perceived to be exactly like others on the market, consumers have no reason to buy it. The effective positioning begins with actually differentiating the company’s marketing offer so gives consumers more value.
Marketing strategies for competitive advantage
Before designing the competitive strategies the company need through analysis of their competitors. In this case the company. Four types of strategies available for marketers, they are-
- Market Leader Strategies.
- Market Challenger Strategies.
- Market Follower’s Strategies.
- Marker Niche Strategies.
Developing the market mix
Marketing Mix, factors that help a company or firm sell its products. Four elements are normally distinguished: getting the right product to the market; selling the product at the right price; ensuring that the promotion is right—that is, advertising and marketing for the product; and ensuring that the product is distributed to the most convenient place for customers to buy it.
On the basis of the marketing strategies the marketer decides the appropriate combination of the marketing mix. Marketing response it wants in the target market. The marketing mix consists of everything the firm can do to influence the demand for its products. The four main tools of the marketing mix from the seller point of view are 4Ps- product, price, place and promotion. From the buyer point of view it includes – customers cost, convenience and communication. Now-a-days marketers consider packaging as new tool of marketing mix and consider it as four Ps.
Product
Means the goods and services that the company offers to the target market to satisfy their states or in implied needs.
Price
Price means the amount of money that the customer has to pay to obtain the product.
Place
Place includes those activities that make the product available to target customers. Place doesn’t main only the market place. It also includes the activities such as : Channel, coverage, assortment, location, transportation and so on.
Promotion
Promotions include the activities that the marketer used to communicate and inform their customer about the products. There are some tools of promotion. These tools are also known as communication mix or promotion mix. They are: Advertising, personal selling, public relation and sales promotion. So to be succeeding the marketer must make proper blend of the marketing mix.
Managing the marketing effort
The company wants to achieve its marketing objectives is its target market. And for this marketer need to manage all the efforts with proper analysis planning, implementation and control.
Marketing analysis
To manage the marketing functions the marketer needs to analyze the company situation. For this the marketer needs to conduct the SWOT analysis. In this case the SWOT means (S- strength, W- Weakness, O- opportunities, T- threats.). Here the strength, weakness are the internal analysis and opportunities, threat are external analysis of the company.
Marketing planning
Marketing planning involves deciding on marketing strategies that will help the company attains its overall strategies objectives. A detail marketing plan also need for all company, as product or brand. Marketing plan includes the following eight elements.
- Executive Summary
- Currant Market Situation
- Threats And Opportunities Analysis
- Objectives And Issues
- Marketing Strategies
- Action Program
- Budgets
- Controls
Marketing implementation
Marketing implementation is the process that turns marketing plans into marketing action in order to accomplish strategies marketing objectives. Marketing planning addresses what and why of marketing activities, implementation addresses who, where, when and how.
Many manager thinks that “doing things right” (implementation) is as important as or even more important than “doing the right things” (strategy). The fact is that both are critical to success.
Marketing department organization: modern marketing department can be arranged in several ways, are-
a. Financial organization
In this type of organization, a functional specialist heads different marketing activities, such as sales manager, advertising manager, customer service, manager.
b. Geographic organization
In this type of organization, sales and marketing people assigned to specific countries, region and district .The company who sales across the country or internationally uses this type of design.
c. Product management organization
In this type of organization, there are some managers responsible for different product or brands. A product, manager develops and implements a complete strategy and marketing programs for a specific product or brands.
Marketing control
To get the proper result the marketer need to measure whether their objectives are fulfilled or not. And for this purpose the marketer takes the initiative that is known as control. Marketing control is the process of measuring and evaluating the result of marketing strategies sand plans and taking corrective action to measure the objective are achieved. It involves the four steps-
Step: 01 set goal
Step: 02 measures performance
Step: 03 evaluate performance
Step: 04 take corrective action.
Management takes the corrective action to close the gap between its goal and its performance. There are some available for the marketer to control their activities. One of the major tools is marketing audit.
Marketing audit
Marketing audit is comprehensive, systematic, periodic and independent examination of a company’s environment, objectives, strategies, and activities to determine the problem areas and opportunities and to recommend plan of action to improve the company’s marketing performance. The audit provides good input for a plan of action to improve the company marketing performance. There are six main elements of marketing audit. They are as follows-
- Marketing environment audit
- Marketing strategy audit
- Marketing organization audit
- Marketing system audit
- Marketing productivity audit
- Marketing function audit
Marketing and Internet
Marketing strategy in the new digital age-
- E-business
- E-commerce
- E-purchasing
- E-marketing
Setting up an e-commerce presence-
Companies can conduct e-marketing in 4 ways. They are-.
- Creating web site
Designing an attractive web site considering 7 C’s
- Context
- Content
- Community
- Customization
- Communication
- Connection
- Commerc
- Placing adds and promotions on line, of different types
- Skyscrapers
- Rectangles
- Interstitial
- Sponsor of browser adds
- Content Sponsorship
- Setting up a web community.
- Using e-mail or web casting.
Pacific Bangladesh Telecom Limited
CDMA and Mobile phone introducer in Bangladesh
CDMA
Code Division Multiple Access (CDMA) is a radically new concept in wireless communications. It has gained widespread international acceptance by cellular radio system operators as an upgrade that will dramatically increase both system capacity and the service quality.
CDMA is a form of spread-spectrum, a family of digital communication techniques that have been used in military applications for many years. The core principle of spread spectrum is the use of noise-like carrier waves, and, as the name implies, bandwidths much wider than that required for simple point-to-point communication at the same data rate.
One of the most important concepts to any cellular telephone system is that of “multiple access”, meaning that multiple, simultaneous users can be supported. In other words, a large number of users share a common pool of radio channels and any user can gain access to any channel (each user is not always assigned to the same channel). A channel can be thought of as merely a portion of the limited radio resource, which is temporarily allocated for a specific purpose, such as someone’s phone call. A multiple access method is a definition of how the radio spectrum is divided into channels and how channels are allocated to the many users of the system.
Benefits of CDMA
The CDMA technology gives you certain benefits over the other prevalent mobile technologies some of them are-
- Superior voice quality and clarity by eliminating background noise.
- Low power consumption, hence higher talk time, enhanced battery life.
- Minimum health risks compared to other wireless technologies.
- Increased security and privacy as none other then the called person can listen to your conversation.
- Higher data transfer rates.
- Reduced interference on other electronic devices.
Company Profile
Name
Pacific Bangladesh Telecom Limited (since 1993)
8 For building appropriate and leading edge Technologies.
8 Cellular and fixed wireless services
8 Offices in Dhaka, Chittagong & Sylhet
Mission Statement
“Because we care“
(To connect people digitally)
Brand Name
CityCell Digital
Service
Pacific Bangladesh Telecom Limited is the first company in Bangladesh to provide mobile telecommunication service and has constantly given its customer more that one reason to be proud of, while the innovation & up gradation in network has resulted in the lowest fault rate in the country. CityCell’s sales & customer service team has won many hearts through their efficiency, skill & customer focus attitude.
Achievements
- Pacific Bangladesh Telecom Limited has consistently set the benchmark of the telecom industry in Bangladesh-
- First to launch Mobile phone in the Sub-continent
- First to adopt CDMA technology in the sub-continent.
- First ever private wireless network in Bangladesh.
- First to connect Cox’s Bazaar to rest of the world.
Pacific Bangladesh Telecom Limited technological up gradation has constantly been setting standard for other service provider in the country. We upgraded our network from the AMPS to the state of art CDMA technology. Now we can boast of the SDH microwave network connecting area between Dhaka – Chittagong and Dhaka – Sylhet.
Total number of employees
Nine Hundreds and Fifty (950) approximately
Total customers
Nine Lakhs and Seventyfive thousands (9.5 lakhs) approximately
Coverage areas
Six (6) Divisions, almost 50 districts & 409 Unions.
Service categories
Prepaid
Alap 24, Alap A, Alap B, Alap Call Me.
Post Paid
Citycell Premium, Citycell 500, Shabar Phone, Amar Phone, Tellular-Tel-To-Cell (CDMA FWT- Fixed Phone)
Main Branch
Pacific Telecom limited
Pacific Centre
14 Mohakhali C/A, Dhaka 1212
CityCell Digital: Their Marketing Strategies
Its true that CityCell Digital was the first company in Bangladesh to provide mobile telecommunication service and was to have a large share in the market, but due to the emerge of other companies like Grameen Phone its facing stiff competition . It’s surprising that the Grameen Phone has taken their share and now is the leader in the market having almost 17 lakhs customers where CityCell is having only nine lakhs and seventy-five thousands.
Taking this painful fact in to consideration, according to the marketing manager the company has adapted many strategies, which not bore satisfactory fruits. To bring a dynamic change in to the market, the company from time to time made many R&D and summing all from past they have developed a new set of strategy. This set of strategies, according to the marketing manager has brought the change, their expected change.
Due to restrictions, in spite of his willingness, the manager could not give us the exact strategies. He did not disappoint us and given some marketing strategies which they follow at the primary level. For this they consider the following things-
¬ The present situation of the company.
¬ Market share of the company.
¬ Financial capabilities of the company to launch a new product and expand network coverage.
¬ Their previous & new product
¬ The special features should be included in the new product.
¬ How different product is from the competitors offerings.
¬ Selecting mobile sets on the basis of market demand, their special features and prices.
¬ Determining the products pricings.
¬ Determine attracting customers on small or large basis.
¬ Consider the economic condition and customer’s affordability, pricings and the competitors’ offerings.
¬ Offer extra benefits to the customers.
¬ Make R&D to find out new facilities and the companies weak ness.
¬ Make R&D to find out what the customers really needs and their present satisfaction.
¬ Let the customers know what the offerings are.
¬ Distributing the products and its methods.
¬ Best timing to launch the product.
¬ Best timing to reduce prices.
¬ Adapt and follow marketing concept
¬ Increase the market share and customers’ satisfaction.
¬ Develop the existing products.
Answers to Some of These Questions
The present situation of the Company
The present situation of the company is much better than the previous one. But it’s still struggling to oust their competitors. Within the last two years they have included many of towns in to their network coverage. Their customers also have increased from around 5 lakhs to almost nine lakhs and seventy-five thousands.
Their Previous & New Product
The products that they offered previously were much expensive and the mobile handsets were much bigger in size. The present products that they are offering are much smaller in size and cheaper too. They are supplying the current demand.
Their Network Coverage
At present they have a comparatively large number of areas under network coverage. They have coverage on 5 divisions –almost 50 districts and 409 unions. They are determined to bring the whole Bangladesh under their coverage within February, 2005.
Special feature and pricings of the Products offered
The main feature of CityCell products is in the facts that they are facilitating the option of T&T incoming and out going in all the categories. In this case they still ahead of the other companies where they are giving T&T incoming and out going only for a single or two categories.
The pricing too is comparatively cheaper. Their products costs from tk.4999 to tk.25999 where as the other companies are offering from tk.5345-tk.39320. The other companies are charging for nationwide roaming and citycell subscribers don’t charge any thing.
Selecting Mobile sets
At the beginning they were offering only Motorola mobile phones but at present due the increased demand of Nokia, Samsung and other ones they had to include them too.
Determining the product prices
- The pricings are determined on the basis of
- How much the mobile set costs.
- Vat and taxes paid to the government.
- Expenditures.
- Commissions to the agents and etc.
Attracting the customers
They don’t try to attract any segment of the market rather they target the market as a whole.
Extra benefits to the customers
They have added a number of extra benefits to the customers. Some of these benefits are free talk time, off peak, super off peak, friends and family numbers, through which the customers talk to any three or two chosen numbers at half rate through out the day.
It should be noted that while CityCell offers 2-3 numbers talking at half rate, to both its pre and post paid customer, other companies are offering 1 for their pre-paid and 3 for their post-paid customer.
Moreover the subscribers can also get information instantly by messaging at a much cheaper cost on train schedules, local and international news, sports news, flight schedule, delicious cuisine, access to police station and fire services.
The latest service that they introduced to the customers is one second pulse facility to the post-paid subscribers which was barred before.
The Customer Care Service
Each subscriber of CityCell can use helpline 121 (from CityCell Mobile) and 011-121121 (from any mobile. Each citycell subscribers can, on arising any problems, call to either *800(free of charge for all prepaid customers) or 011810888 and place their complaints.
More over the customers can also use the online help from www.citycell.com, they can find out each and every thing about their mobile sets, prices and other benefits. They can also place online complaints and get solutions instantly.
Packaging and distributing
When the mobile phones are sold they are not given any extra packaging. The phones are given in the boxes that they get from the mobile companies and in addition the customer gets an attractive medium sized shopping bag in which the whole CityCell Digitals logo is embossed.
The products gets distributed through their won company vehicles and in some cased they take the help of physical distribution firms.
They have a number of agencies and distributors who bye the products from the Pacific Telecom Ltd. and resell them in to the local market. For this transaction they get a certain percentage of commission with some facilities included.
Best time to launch the product
Whatever product they develop, they launch it immediately but they wait for events or festivals such as “ The Baisaki” , ”Eid” or when the new budget or taxation on mobile phones are reduced.
Following marketing concept
According to the marketing manager, the needs and wants of customers never remain same. It changes. Taking this in to consideration the company develops and offers a variety of products with different features. This has been a regular route from time to time. He also says is the customers needs and wants are met they are automatically satisfied and their loyalty gets established or goes high.
Since, each information they give concerns the inside of the company, the marketing manager could mot give us information on questions such as how do they increase the market share, how do they develop the existing products or when and how do they reduce the prices.
CityCell Digital: Their Marketing Activities
According to the marketing manager it’s the marketing activity through which the products are exposed to the customers and each activity should be dynamic and best enough attracting and best enough to bring the customers in and not out. Its just the way a bees is attracted to the flowers.
Just to ensue this in the best possible way not only CityCell Digital but others too spend a good lot of money for advertisements so that the bees are attracted to them. Some of the ways they carry out their marketing activities are mentioned bellow.
Advertisements on
Television Channels
Till now they have given two television advertisements one of 36 seconds and the other is of almost 41 minutes. The models were well known Tisha and Nobel and the jingles were “Dheet Deri Hoey Gelo ………. Raag Koiro na Bondhu amar” and “Nishethe Call Koiro Amar phone”. Among these two advertisements the later one was much successful in their attempt.
Newspapers.
The advertisements on newspapers are almost regularly given by the mobile phone companies. The two key reasons behind this is, its affordable and read in mass by the people. While they choose the newspaper, the highest selling newspaper is always given priority and CityCell puts their advertisements on the best selling and distributed newspapers.
When they place the ads they focuses more or highlights mainly on the items that takes the customers attentions at the first sight. For example- when they advertised for their products pricings the highlights the mobile phones and their prices which are always in figures such as tk.6999 or tk.7999. This is a Psychological Pricing, where the customers take in to account the figure as rounded.
Sponsoring Programs
CityCell sponsors a number of programs. The programs that they sponsor are mainly on televisions channels (part of news’s at 8 and 10), sections of magazines (Binodon), and hot talks on television channels. Their sponsored programs use their logo and let the public see. This in turn also puts an effect in to their minds.
Roadside Billboards
A number of billboards and signboards are seen in Dhaka and outside showing the CityCell logo and a message for public awareness below.
Bringing celebrities – who answers customers questions
This is a newly program undertaken by CityCell, in which it brings superstars who answers customers questions. The customers do respond great. The named it as “CityCell- Tarar Sathe “.
Here nothing but each superstar brought in way or other becomes a model for CityCell and advertises in a way or other for them.
Undertaking quiz programs
Another interesting way of advertisement is sponsoring and undertaking quiz programs. When the SSC result came out, they undertook such program. This was published in the Daily Prothom Alo and a huge numbers of students participated.
There are some other activities to be taken by the company. But what are they were not disclosed to us remains just a sort of surprise.
Facility for Subscriber
Cash Card
Cash card is a type of scratch card, which allows post-paid customers to pay their phone bills through their own CityCell mobile.
Benefits
- Ø Customers don’t have to wait for the bill statement.
- Ø Cash card allows customers to avoid disconnection by making advance payment from anywhere, anytime.
- Ø Customers can avoid waiting in a long queue in the bank.
- Ø Customers in the remote area (where banking facility is unavailable) can get uninterrupted service.
ü Voice Mail Service (VMS)
ü Short message Service (SMS)
ü Nationwide Roaming Facility
ü International Roaming Facility
ü Twenty four hours computerized Customer Service.
ü Itemized Bill
ü Calling Line Identification Presentation (CLIP)
ü Call Forwarding \ Diverting
ü Call Waiting \ Holding
ü Call Barring
ü Help Line
Each subscriber of CityCell can use helpline 121 (from CityCell Mobile) and 011-121121 (from any mobile.
Property Status
The company is owned and finance by two organization one is Pacific Bangladesh Telecom Limited and another one is Singapore Telecommunications Limited (singtel) and their share on the organization is-
First Initiatives
ü First time introduce mobile phone in Bangladesh. The first launched mobile company in Bangladesh.
ü First time CDMA mobile service introducer in Bangladesh.
ü They have introduced the SMS facility with other mobile phone service organization of Bangladesh.
ü They are the first introducer of Friends and family number in all the mobile phone service companies of Bangladesh.
ü First time introducer of one second pulse in a package for the better facility of the subscribers.
Conclusion
This was not at all an easy project to complete. Gathering information was also a difficult task. What ever the limitations were, we made our best effort to overcome them and anchor safe.
No matter how good we do in designing the project but it’s the knowledge that we gather that remains us till the end and satisfies.At last, we hope that our attempt behind this project is a successful one and will surely bear the expected fruit.
References
- http://www.citycell.com
- Principles of Marketing
Philip Kotler
Gray Armstrong
- Fundamental of Marketing
William J Stanton
Michael Etzel
Bruce J Walker
Questionnaire
♣ What are the strategies that you follow?
♣ What is your marketing planning?
♣ How and when do you reduce the rates?
♣ Why do you use CDMA phones and not the GSM ones?
♣ What are the main ideas behind giving advertisements?
♣ How are the products supplied?
♣ What are the marketing activities?
♣ How are the prices fixed?
♣ How are the customers selected?
♣ Why is market share so less?
♣ What are the areas under your coverage?
♣ Does all you plan and undertake help to reach your goals?
♣ What is your total customer?
♣ Do you have any market segment?
♣ How do you plan out the new products?
♣ What do you do for your customer care / satisfaction?
♣ What are your future planning’s?
♣ How do you make the R&D?
♣ How are the goods selected?
♣ Who are your main competitors?
♣ Do you think you put a competition to the competitors?
♣ Are the competitors putting any competition on you?
♣ How do you choose the repeated customers?
♣ How much is your expenses for advertisements?
♣ How do you maintain the prices after giving such additional products?