Calcite is a carbonate mineral and the most stable polymorph of calcium carbonate (CaCO3). The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 3 as “calcite”. It was named after a Latin word ‘calx’, meaning burnt lime, which is an allusion to one of its important commercial uses.
Other polymorphs of calcium carbonate are the minerals aragonite and vaterite. Aragonite will change to calcite at 380–470 °C and vaterite is even less stable.
General Information
- Category: Carbonate minerals
- Formula: CaCO3
- Crystal system: Trigonal
- Crystal class: Hexagonal scalenohedral (3m)

Properties
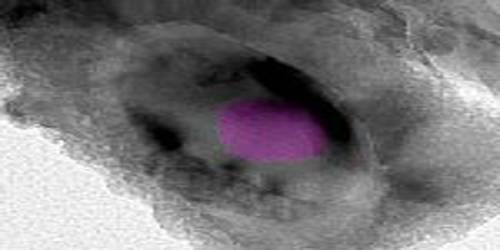
Calcite can be identified in the field by its color variations, such as white, pink, yellow, and brown. It is sometimes colorless. This mineral has a vitreous luster with a white streak. The fracture on this mineral is brittle – conchoidal. The density of calcite is 2.71 g/cm3, with a hardness of 3 – approximate to calcite. It is fluorescent and phosphorescent.
- Color: Colorless or white, also gray, yellow, green,
- Crystal habit: Crystalline, granular, stalactitic, concretionary, massive, rhombohedral.
- Twinning: Common by four twin laws
- Fracture: Conchoidal
- Tenacity: Brittle
- Mohs scale hardness: 3 (defining mineral)
- Luster: Vitreous to pearly on cleavage surfaces
- Streak: White
- Diaphaneity: Transparent to translucent
- Specific gravity: 2.71
It has a defining Mohs hardness of 3, a specific gravity of 2.71, and its luster is vitreous in crystallized varieties. Color is white or none, though shades of gray, red, orange, yellow, green, blue, violet, brown, or even black can occur when the mineral is charged with impurities.
Occurrence
Calcite occurs in major rock-forming minerals; in limestones, marbles, and chalks; a common cement in clastic sedimentary rocks; as gangue in hydrothermal veins; in alkalic to mafic igneous rocks; and commonly as speleothems in caves.
It is often associated with minerals such as dolomite, Celestine, fluorite, barite, pyrite, marcasite, sphalerite, zeolites, talc, chalcedony, ‘chlorite’, tremolite, grossular, quartz, nepheline, diopside, orthoclase, and apatite.
Information Source;