Anti-Müllerian Hormone
Definition
Anti-Müllerian Hormone (or AMH) is produced by small growing follicles, thus is distinct from ovulation and is a step closer to being able to assess the true ovarian reserve. AMH is measureable from birth to near the menopause, with a peak in the mid-20s. It is a glycoprotein hormone from the transforming growth factor beta superfamily. Encoded by the AMH gene, its primary known roles are in growth differentiation and folliculogenesis.
Anti-Mullerian Hormone (AMH) helps to form the male and female reproductive organs. It also helps control other hormones that act on the ovaries to make the follicles and eggs grow during a woman’s menstrual cycle. Measuring the amount of anti-Mullerian hormone in the blood may help diagnose infertility and other conditions, such as polycystic ovarian syndrome, menopause, and certain types of ovarian tumors. It may also help determine the sex of an infant, when the sex is not clear.
AMH expression also occurs in ovarian granulosa cells of females postpartum, and serves as a molecular biomarker for relative size of the ovarian reserve. In humans, the number of cells in the follicular reserve can be used to predict timing of menopause. In bovine, AMH can be used for selection of females in multi-ovulatory embryo transfer programs by predicting the number of antral follicles developed to ovulation.
Anti-Mullerian Hormone (AMH) is directly secreted from the preantral and antral follicles, and has the useful property of reflecting the age-induced decrease in the number of follicles. Serum AMH levels are known to be a highly reliable marker for measuring ovarian reserve because they are not affected by gonadotropin, and AMH exhibits minimal variability within or among menstrual cycles. Currently, the presence of a proportional relationship between AMH levels and the ovarian response to COS is commonly accepted. According to recent studies, serum AMH levels have been found to better reflect ovarian reserve than age or basal levels of FSH, estradiol, and inhibin B.
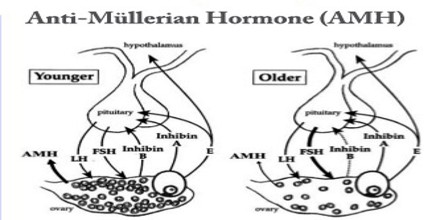
Production of Anti-Mullerian Hormone (AMH)
The key findings are that AMH expression by granulosa cells becomes detectable as soon as follicles start to grow, but later declines. The smallest growing follicles are numerically the most abundant in the ovary, but having few granulosa cells means that they probably contribute little to serum AMH concentrations compared to larger follicles. AMH is expressed in growing follicles throughout the pre-antral and early antral stages, and then declines with little production beyond 8 mm.
Anti-Mullerian Hormone (AMH) production has declined when follicles are selected for dominance and when oestrogen production starts to escalate, and a contribution to this switch may be one of the functions of AMH. Large follicles produce little AMH, expression persists only in the cumulus cells surrounding the oocyte); thus, AMH does not show clinically relevant changes across the menstrual cycle. This greatly adds to its clinical utility, particularly compared to FSH, the historically established maker of ovarian reserve.
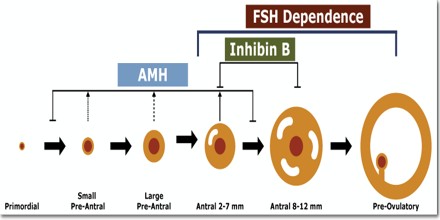
Structure and Function of Anti-Mullerian Hormone (AMH)
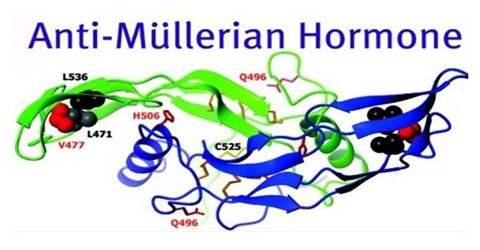
Anti-Müllerian Hormone (or AMH) plays a key role in male sexual differentiation is a glycoprotein, produced by Sertoli cells. It has a molar mass of 140 kDa. In male fetuses AMH is responsible for regression of Müllerian duct, the analgen of uterus, the Fallopian tubes, and 2/3 upper of the vagina, while in female fetuses for a decrease in aromatase activity in granulosa cells of the ovary. Human AMH gene consists of 5 exons: 412, 124, 108, 160 and 856 bp in length, and is localized near the tip of chromosome 19, in subbands 13.2 to 13.3. Mutations of the gene or the receptor of AMH are responsible for clinical symptoms of Persistent Müllerian Duct Syndrome (PMDS) which occurs in subjects with a karyotype 46XY and a male phenotype. Typical case is that of a male with bilateral cryptorchidism and inguinal hernias but normal male genitalia. Uterus and Fallopian tubes are often present in the inguinal canal.
Anti-Müllerian Hormone (AMH) is expressed by granulosa cells of the ovary during the reproductive years, and limits the formation of primary follicles by inhibiting excessive follicular recruitment by FSH. Some authorities suggest it is a measure of certain aspects of ovarian function, useful in assessing conditions such as polycystic ovary syndrome and premature ovarian failure. It is useful to predict a poor ovarian response in in vitro fertilization (IVF), but it does not appear to add any predictive information about success rates of an already established pregnancy after IVF. Additionally, AMH levels are used to determine a women’s remaining egg supply.
Anti-Müllerian Hormone (AMH) in Diagnosis
Anti-Müllerian Hormone (AMH) may also have a role in the diagnosis of ovarian dysfunction. Its relationship with some of the common causes of oligo/amenorrhoea has been discussed above, but its clearest potential is in the diagnosis of polycystic ovarian syndrome (PCOS). Women with PCOS often have very markedly increased AMH concentrations. The high intra-ovarian AMH concentrations may also contribute to a reduced responsiveness to FSH. Measurement of AMH may be of value in the differential diagnosis of oligomenorrhea, and it is likely that it will be part of future revisions of the criteria for the diagnosis of PCOS.
Anti-Müllerian Hormone (AMH) is also of value in the diagnosis and monitoring of granulosa cell tumours, and the finding of a very high concentration should lead to consideration of this diagnosis. The production of AMH by the testicular Sertoli cell underlies its emerging value in the differential diagnosis of disorders of sexual development.