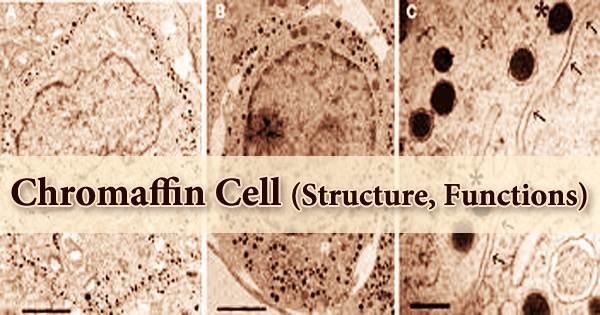
The proliferation of chromaffin cells, also known as pheochromocytes, in the rat adrenal medulla rapidly decreases after birth. In animals, these neuroendocrine cells are usually found in the medulla of the adrenal glands. These cells perform a range of activities, including stress response, monitoring carbon dioxide, and oxygen concentrations in the body, respiration maintenance, and blood pressure management. Mitoses are easily visible in postnatal rat adrenal histological sections, although they are uncommon after 60 days. For many years, adult rat chromaffin cells were thought to be essentially postmitotic and terminally differentiated.
The hypothalamic-pituitary adrenocortical axis and the autonomic nervous system are both regulated by corticotropin-releasing hormone (CRH). Following research, it was shown that E and NE cells multiply throughout life. They connect with pre-synaptic sympathetic ganglia of the sympathetic nervous system, and they have a structure that is similar to that of post-synaptic sympathetic neurons. Mitoses are exceedingly rare in SGC cells, and there is no evidence that SGC cells are progenitors to conventional E or NE cells.
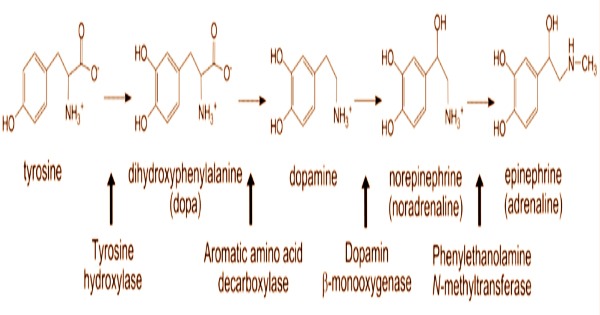
The sympathetic nervous system’s splanchnic nerve releases acetylcholine, which binds to nicotinic acetylcholine receptors on the adrenal medulla to activate chromaffin cells. This causes the release of catecholamines. Adrenocortical steroids impact the structure and function of the chromaffin cells in the adrenal medulla, while catecholamines and neuropeptides produced in the adrenal medulla govern adrenocortical function. The chromaffin cells release catecholamines into the systemic circulation, including ~80% of adrenaline (epinephrine) and ~20% of noradrenaline (norepinephrine), which have systemic effects on many organs and can also send paracrine signals.
Chromaffin cells are formed from the neural crest of the human embryo. The sympathetic ganglia, vagus nerve, paraganglia, and carotid arteries are all near them. They’re located in the bladder wall, prostate, and behind the liver, to name a few places. A short course of administration of reserpine, one of the many drugs that generate adrenal medullary tumors following chronic administration, has been found to increase chromaffin cell proliferation in adult rats.
There are two types of neural crest cells that are associated to the sympathetic nervous system (they come from a cell called sympathogonia)
- Neuroblasts: These cells migrate on both sides of the spinal cord toward the region directly behind the dorsal aorta during the fourth to fifth week of fetal development in humans, forming the two sympathetic ganglia chains (Sympathetic chain). Some of these cells will move to the adrenal medulla, where they will produce sympathetic ganglia cells (without postsynaptic sympathetic fibers).
- Chromaffin cells (or pheochromocytes): These cells will move to the adrenal medulla and the area next to the sympathetic ganglia (thus the name paraganglia). The organ of Zuckerkandl is the largest extra-adrenal cluster of chromaffin cells in mammals. The vagus nerve and carotid arteries are also home to chromaffin cells. Extra-adrenal chromaffin cells can also be found in the bladder wall, prostate, and behind the liver at lower numbers.
In response to increased physiological demands, the mitogenic impact could theoretically serve to raise the functional reserve of the adrenal medulla. The proliferation of rat chromaffin cells in cell culture was stimulated by agents that mimicked brain stimulation by activating intracellular signaling pathways known to mediate the actions of acetylcholine or peptide neurotransmitters. In non-mammals, chromaffin cells can be located in a variety of locations, are rarely structured as a separate organ, and may lack innervation, relying only on endocrine or paracrine cues for secretion.
The splanchnic nerve innervates chromaffin cells in the adrenal medulla, which release adrenaline (epinephrine), noradrenaline (norepinephrine), some dopamine, enkephalin and enkephalin-containing peptides, and a few other hormones into the bloodstream. Control adrenomedullary cells displayed the typical ultrastructural features of neuroendocrine cells, including an abundance of membrane bound secretory spanning in size from 60 to 400 nm in maximum dimension, and rough endoplasmic reticulum (RER).
Endogenous peptides known as endorphins (secreted from the pituitary) are related to, but separate from, enkephalins and enkephalin-containing peptides; all of these peptides bind to opioid receptors and produce analgesic (and other) reactions. Epinephrine and norepinephrine were the two main forms of chromaffin vesicles. Medium-density vesicles carrying epinephrine were big, circular, or elongated. The vesicles carrying norepinephrine were tiny and electron-dense.
Similarly, an increase in liposomes, which are cholesterol storage organelles, indicates diminished steroid production and a hypofunctional adrenocortical condition. The hormones are produced in chromaffin granules, which contain the enzyme dopamine β-hydroxylase, which catalyzes the conversion of dopamine to noradrenaline. In this symposium, the chromaffin cell was highlighted as a significant neuroendocrine depot from which epinephrine is released during both systemic and psychological stress responses.
Pheochromocytomas are sometimes used to refer to just adrenal neoplasms, while extra-adrenal paragangliomas are used to refer to extra-adrenal paragangliomas. The sympathetic nervous systems (SNS) principal output is chromaffin cells in the adrenal medulla. Individual chromaffin cells are excited to perform exocytosis when action potentials are fired via cholinergic synaptic input from the sympathetic splanchnic nerve. Pheochromocytes are another name for chromaffin cells. Although enterochromaffin cells have a histological resemblance to chromaffin cells, their function is fundamentally different, and they are not neural crest descendants.
Information Sources: