Scientists have developed a novel strategy for reducing Alzheimer’s disease (AD) pathologies in genetically modified AD mouse models using brain-wide genome-editing technology. This cutting-edge technology has enormous potential to be translated into a novel long-acting therapeutic treatment for Alzheimer’s disease patients.
An international research team led by scientists from the Hong Kong University of Science and Technology (HKUST) has developed a novel strategy for reducing Alzheimer’s disease (AD) pathologies in genetically modified AD mouse models using brain-wide genome-editing technology. This cutting-edge technology has enormous potential to be translated into a novel long-acting therapeutic treatment for Alzheimer’s disease patients.
Over 500,000 patients are estimated to be living with a hereditary form of Alzheimer’s disease (FAD), which is a congenital form of Alzheimer’s disease strongly linked to family history, in China alone. Although FAD has a clear genetic cause and can be diagnosed before cognitive problems arise, there is currently no effective treatment.
Genome editing is a technology that precisely modifies the genomic DNA of a living organism by deleting, inserting, or replacing DNA at specific locations on the genome.
Scientists have developed a novel strategy using brain-wide genome-editing technology that can reduce Alzheimer’s disease (AD) pathologies in genetically modified AD mouse models.
The use of genome-editing technology as therapeutic strategies for diseases caused by inherited mutations, such as FAD, has enormous potential. It is especially useful for correcting disease-causing genetic mutations before symptoms appear, and its effects can last a lifetime, making it a “once-and-for-all” treatment. However, several obstacles have stymied its clinical development and application, the most significant of which is the lack of an effective, efficient, and non-invasive method of delivering genome-editing agents into the brain. Furthermore, current genome-editing technologies are incapable of producing beneficial outcomes across the entire brain.
Prof. Nancy Ip, Vice-President for Research and Development at HKUST, recently led a team that developed a new genome-editing system that not only crosses the blood-brain barrier but also delivers an optimized genome-editing tool to the entire brain. This strategy achieves efficient brain-wide genome editing through a single non-invasive intravenous administration using a newly engineered genome-editing delivery vehicle. This effectively disrupts FAD-induced mutations in AD mouse models and improves AD pathologies across the entire brain, paving the way for novel therapeutic development for the disease.
Meanwhile, the researchers discovered that the level of amyloid, a protein thought to drive neurodegeneration in Alzheimer’s disease, remained low in the mouse models for 6 months after treatment (roughly 1/3 of their normal lifespan), demonstrating that this single-shot genome-editing strategy has long-term effects. More importantly, no side effects have been observed in the mice thus far.
“This is a really exciting development as the first demonstration of efficient brain-wide genome editing to alleviate Alzheimer’s disease pathology throughout the entire brain,” said Prof. Ip, who is also the Morningside Professor of Life Science and Director of the State Key Laboratory of Molecular Neuroscience at HKUST. “Our work represents an important milestone in the use of genome editing in the treatment of hereditary brain diseases, and it contributes to the development of precision medicine for inherited forms of neurodegenerative disease.”
Scientists from HKUST, the California Institute of Technology, and the Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences collaborated on this study. The findings were recently published in the journal Nature Biomedical Engineering.
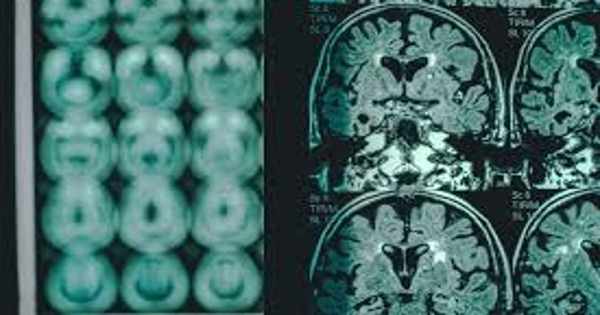
Clinically, patients with Alzheimer’s disease most commonly present with insidiously progressive memory loss, which is accompanied by impairments in other areas of cognition over time. In addition to memory loss, patients may experience language difficulties (e.g., anomic aphasia) and loss of executive skills, all of which are symptoms of dementia.
The pathophysiology of Alzheimer’s disease (AD) is known to be linked to gene mutations, specifically presenilin (PSEN) and amyloid-beta precursor protein (ABP) (APP). Clinical trials focusing on the genetic level, on the other hand, we’re unable to achieve the desired efficiency. The CRISPR/Cas9 genome editing tool is one of the most powerful technologies for correcting inconsistent genetic signatures and is now widely used in the treatment of Alzheimer’s disease.
AD is a common type of dementia that affects the health of millions of people worldwide. Despite the fact that it has been known for over a century, numerous questions about its pathophysiology remain unanswered. Decreased cognitive abilities, such as memory, recognition, judgment, and problem solving, are typical clinical features of Alzheimer’s disease.