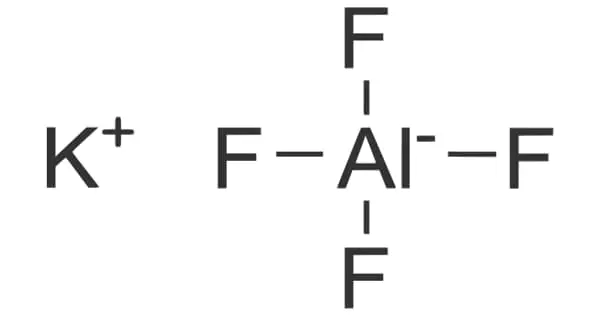
Potassium aluminium fluoride (KAlF4) is an inorganic compound with the chemical formula KAlF4. It is a key component in salt fluxes for metal cleaning, along with other chloride and sulphate salts such as NaCl. The addition of fluoride salts improves the salt flux’s performance. These salt fluxes aid in the separation of metals from their oxides and dross, as well as preventing further burning of the molten aluminum, increasing its yield.
This compound is used as a flux in the secondary aluminum smelting process to reduce or remove the magnesium content of the melt. The main environmental issue caused by PAF use is the production of fluoride gases. Calcium hydroxide is widely used to suppress fluoride production, but it rarely removes it completely.
Potassium Aluminum Fluoride is a fused inorganic salt of commercial purity. It possesses beneficial properties and is used in a variety of industrial applications. The compound is used as a salt flux in secondary aluminum smelting to maximize metal recovery, to reduce or remove the magnesium content of liquid aluminum alloys, and as a filler in abrasive manufacturing. PAF is also used as a fluxing agent in metal additives to speed up dispersion after addition.

To improve product performance, PAF is used as an active filler, and/or an active topcoat ingredient is bonded (primarily resin bonded) and coated abrasives. The use of active ingredients is increasing; their impact on abrasive grinding performance is significant. According to one flap disc manufacturer, the use of active substances allows him to either improve the properties of his product or maintain its performance while saving up to 65 percent on the amount of abrasive powder.
Occurrences
A single natural occurrence of an unnamed mineral has been reported at a burning coal bank in Forestville, Pennsylvania. Because PAF is a better heat conductor than Cryolite, is generally less expensive, and is not classified as a dangerous good – resulting in fewer issues in transport, processing, and storage – it is easy to see why its use in various industrial applications, such as the abrasives industry, is still expanding.
Uses
PAF is also found in a variety of metals industry products as a fluxing agent within additives to aid in its dispersion within a charge. It’s also used to kill insects. It is also used directly in magnesium-rich aluminum alloys to remove and control the magnesium content of the alloys.
PAF is also commonly used as an additive to roofing shingles, where it acts as an alkaline Ph adjuster and flame retardant. It can also be used as a color additive in roofing granule coatings.