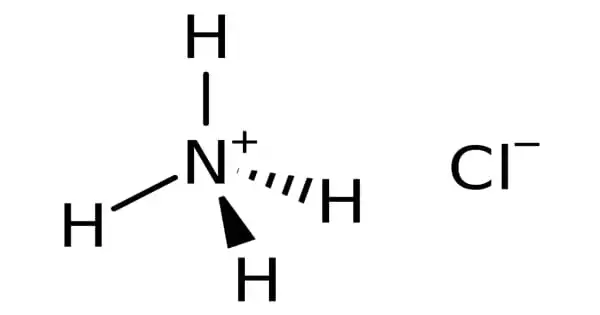
Sodium carbonate produces ammonium chloride as a byproduct. It is a white crystalline salt that is extremely soluble in water and an inorganic chemical with the formula NH4Cl. It is white crystalline salt in its purest form. Ammonium chloride solutions are moderately acidic. In veterinary medicine, it is used to avoid urinary stones in sheep, goats, and cattle.
It is known as salt ammoniac in its naturally occurring mineralogic form. Condensation of coal-derived gases frequently forms the mineral on burning coal heaps. It can also be found near some types of volcanic vents. It is mostly used as a fertilizer and as a flavoring component in certain varieties of liquorice. It is the result of a reaction between hydrochloric acid and ammonia.
Properties
Ammonium chloride is a crystalline white substance. It dissolves in water (37 percent). The primary risk is the threat to the environment. It is a urinary and systemic acidifying salt. Ammonium chloride aids in pH maintenance and has a minor diuretic impact.
- Molecular Weight/ Molar Mass: 53.491 g/mol
- Density: 1.53 g/cm³
- Boiling Point: 520 °C
- Melting Point: 338 °C

Production
It is a product of the Solvay process used to produce sodium carbonate:
CO2 + 2 NH3 + 2 NaCl + H2O → 2 NH4Cl + Na2CO3
Not only is this the primary method for producing ammonium chloride, but it is also utilized to reduce ammonia release in several industrial processes.
Commercially, ammonium chloride is made by mixing ammonia (NH3) with either hydrogen chloride (gas) or hydrochloric acid (water solution):
NH + HCl → NH4Cl
Ammonium chloride is found naturally in volcanic areas, where it forms on volcanic rocks near fume-emitting vents (fumaroles). The crystals form straight from the gaseous state and have a brief lifetime since they disintegrate rapidly in water.
Reactions
Ammonium chloride is an acidifying salt that can be found in the body and urine. It aids in pH regulation and has a minor diuretic effect. This acid-forming salt also possesses expectorant properties by irritating the mucous membranes, making it effective for cough alleviation. Ammonium chloride is a crystalline white chemical.
After cooling, ammonium chloride pyrolyzes and reforms to generate ammonium chloride smoke. It looks to sublime when heated, but it actually decomposes into ammonia and hydrogen chloride gas.
NH4Cl → NH3 + HCl
Ammonium chloride reacts with a strong base, like sodium hydroxide, to release ammonia gas:
NH4Cl + NaOH → NH3 + NaCl + H2O
Similarly, ammonium chloride also reacts with alkali metal carbonates at elevated temperatures, giving ammonia and alkali metal chloride:
2 NH4Cl + Na2CO3 → 2 NaCl + CO2 + H2O + 2 NH3
A 5% by weight solution of ammonium chloride in water has a pH in the range 4.6 to 6.0.
Some of ammonium chloride’s reactions with other chemicals are endothermic like its reaction with barium hydroxide and its dissolving in water.
Applications
The most common application of ammonium chloride is as a nitrogen source in fertilizers such as chloroammonium phosphate (which accounts for 90 percent of global ammonium chloride output). In Asia, the principal crops fed this manner are rice and wheat.
In the 18th century, ammonium chloride was utilized in pyrotechnics, but it was eventually replaced with safer, less hygroscopic compounds. Its function was to act as a chlorine donor, enhancing the green and blue colors produced by the copper ions in the flame. It had a secondary application as a white smoke producer, but its ready double decomposition reaction with potassium chlorate produced the highly unstable ammonium chlorate, making its use extremely hazardous.
Health Effects
Overdosing is a possibility, therefore it’s not fully risk-free. Ammonium chloride can produce an increase in blood pressure. Ammonium chloride poisoning causes irritation, shortness of breath, coughing, nausea, and headache. The gases have the potential to cause severe eye irritation. Chronic exposure can cause asthma-like symptoms or damage kidney function.