The ionic concentrations in the settings where marine fish reside are very different from those in their blood plasma. If a fish does not excrete the hazardous ion species in seawater, they can accumulate in the body.
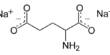
One illustration of this is the vitamin boric acid, which is essential to animals in tiny doses but can be hazardous in large levels. As a result, marine fish need to develop physiological mechanisms of excreting borate.
However, how they do this is, as yet, unknown. The molecular mechanisms underpinning the secretion of boric acid by marine pufferfish have now been revealed and demonstrated by an international team headed by scientists from Tokyo Institute of Technology (Tokyo Tech).
Associate Professor Akira Kato of Tokyo Tech is the principal author of the study, which was published in the Journal of Biological Chemistry. He tells us more about it.
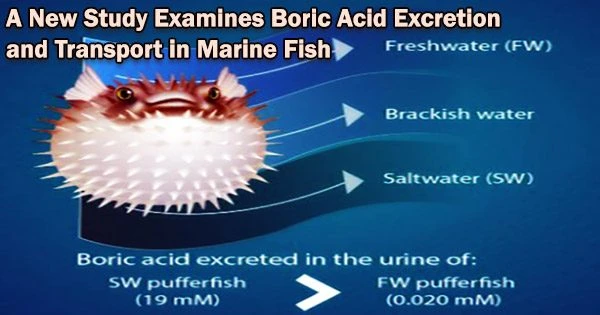
“We compared euryhaline pufferfish (which are pufferfish that can survive in varying levels of salinity) accustomed to saltwater, brackish water, and freshwater. On comparing fish from these three habitats, we found that the urine of a seawater pufferfish (Takifugu pufferfish) contained 300 times more boric acid than pufferfish blood, and 60 times more boric acid than seawater.”
The urine of freshwater fish contained almost 1000 times less boric acid than that of seawater pufferfish. These findings established that Takifugu pufferfish living in seawater excrete boric acid in their urine. Just like in humans, the process of excretion via urine in pufferfish is mediated by the kidneys.
We compared euryhaline pufferfish (which are pufferfish that can survive in varying levels of salinity) accustomed to saltwater, brackish water, and freshwater. On comparing fish from these three habitats, we found that the urine of a seawater pufferfish (Takifugu pufferfish) contained 300 times more boric acid than pufferfish blood, and 60 times more boric acid than seawater.
Associate Professor Akira Kato
How did this boric acid enter the kidney tubules, though?
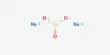
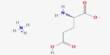
The team found that Takifugu pufferfish expressed an uncharacterized gene, Slc4a11A, in their kidney tubules. This gene encodes a protein homologous to BOR1, which is a boric acid transporter found in plants.
“A detailed electrophysiological analyses of the pufferfish Slc4a11A function revealed that Slc4a11A functions as an active or electrogenic boric acid transporter,” further discusses Dr. Kato.
In essence, this suggests that Slc4a11A’s ability to transport boric acid across a gradient of concentration is independent of other ions like sodium. The fact that this is the first account of an active boric acid transport system in an animal species makes it particularly significant.
Since mammals also possess the Slc4a11 gene, it has been unclear whether or not they are likewise capable of carrying out this job. Human mutations in the Slc4a11 gene result in vision abnormalities such congenital hereditary endothelium corneal dystrophy and Fuchs’ endothelial corneal dystrophy, despite the fact that mammalian Slc4a11 does not transport boric acid.
Further investigation of the functions of Slc4a11 in other vertebrate species will reveal whether mammalian Slc4a11 lost its ability to transport boric acid during evolution or whether ocean fish Slc4a11A developed it.
In any case, this ground-breaking finding creates new avenues for understanding animal boric acid transport and unraveling genetic secrets.