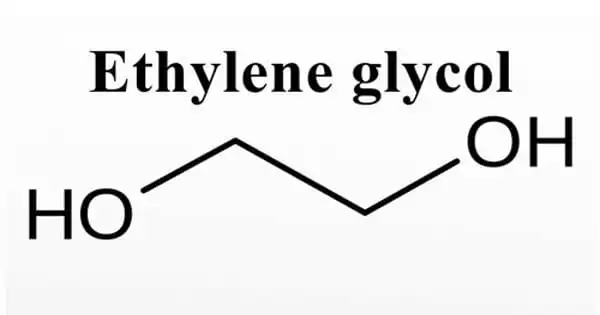
The organic compound ethylene glycol has the formula (CH2OH)2. It is a useful industrial compound that can be found in a variety of consumer products. It is primarily used for two purposes: as a raw material in the production of polyester fibers and in the formulation of antifreeze solutions. Antifreeze, hydraulic brake fluids, some stamp pad inks, ballpoint pens, solvents, paints, plastics, films, and cosmetics are a few examples. It is a toxic, odorless, colorless, sweet-tasting liquid with a viscous consistency.
Ethylene glycol has a sweet taste and is frequently consumed inadvertently or on purpose. In the body, it degrades into toxic compounds. Its toxic byproducts first affect the CNS, then the heart, and finally the kidneys.
Properties
Ethylene glycol is a clear, sweet, slightly viscous liquid that boils at 198 degrees Celsius (388.4 degrees Fahrenheit). Its most common application is as an antifreeze in automobiles. It is a significant organic compound and chemical intermediate that is used in a wide range of industrial processes (e.g. energy, plastics, automobiles, and chemicals). It is highly soluble in water, polar solvents, and insoluble in non-polar solvents. Because of intermolecular hydrogen bonding, the boiling point is extremely high.
Production
Ethylene glycol is created by converting ethylene (ethene) through the intermediate ethylene oxide. According to the chemical equation, ethylene oxide reacts with water to produce ethylene glycol:
C2H4O + H2O → HO−CH2CH2−OH
This reaction can be catalyzed by acids or bases, or it can occur at neutral pH at high temperatures. The highest ethylene glycol yields are obtained when the pH is acidic or neutral and there is a large excess of water. Under these conditions, ethylene glycol yields of up to 90% are possible. The oligomers diethylene glycol, triethylene glycol, and tetraethylene glycol are the most important byproducts. The separation of these oligomers and water requires a lot of energy. Each year, approximately 6.7 million tonnes are produced.
Shell’s OMEGA process is used to achieve higher selectivity. In the OMEGA process, ethylene oxide is first converted to ethylene carbonate by combining it with carbon dioxide (CO2). In a second step, this ring is hydrolyzed with a base catalyst to produce mono-ethylene glycol with 98 percent selectivity. In this step, carbon dioxide is released once more and can be recycled into the processing circuit. The carbon dioxide is produced in part by the ethylene oxide process, in which a portion of the ethylene is completely oxidized.
Toxicity
When ingested, ethylene glycol has high mammalian toxicity, roughly comparable to methanol, with an oral LDLo = 786 mg/kg for humans. The main danger is that its sweet taste attracts children and animals. When ethylene glycol is consumed, it is oxidized to glycolic acid, which is then oxidized to oxalic acid, which is toxic. It and its toxic byproducts attack the central nervous system first, then the heart, and finally the kidneys.
There are antifreeze products for automotive use that use propylene glycol instead of ethylene glycol. They are generally thought to be safer to use because propylene glycol isn’t as tasty and is converted in the body to lactic acid, a normal byproduct of metabolism and exercise.