Ammonia (chemical formula: NH3) is a nitrogen-hydrogen compound. It is extremely soluble in water as a colorless smelly gas, resulting in liquid ammonia. Ammonia is a chemical compound made up of three hydrogen atoms connected to one central nitrogen atom.
Both are relatively common elements; nitrogen makes up the majority of the Earth’s atmosphere, and hydrogen is the most plentiful element in the universe. Ammonia is a nitrogen-containing chemical that is found in abundance in the atmosphere. It serves as a precursor of food and fertilizer for a variety of species, meeting their basic nutrient requirements.
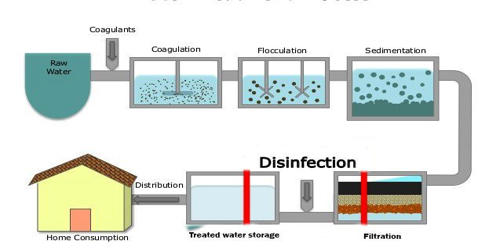
Green ammonia production refers to ammonia production that is 100 percent renewable and carbon-free. Using hydrogen from water electrolysis and nitrogen extracted from the air is one method of producing green ammonia. These are then put into the Haber process (also known as the Haber-Bosch process), which is entirely powered by renewable energy.
In any case, Haber-Bosch and other industrial-scale manufacturing methods necessitate high temperatures (above 400°C) and high pressures (more than 150 bar). To break the strong bonds in nitrogen and react with hydrogen to generate ammonia, certain conditions are required (NH3). It’s also necessary for a variety of biological processes.
Furthermore, it is used to make a variety of commercially essential nitrogen compounds as well as therapeutic goods. Ammonia is an extremely stinky gas having a boiling point of 33.3 °C (28.0 °F) at atmospheric pressure. It’s relatively easy to liquefy when kept cold or under a small amount of pressure, making it a far easier green fuel to transport and store than hydrogen.
The production of ammonia, on the other hand, is currently not a “green” process. It’s most usually manufactured from methane, water, and air, with the hydrogen produced using steam methane reforming (SMR) and the Haber process.
The SMR method produces around 90% of the carbon dioxide produced. This process uses a lot of energy and emits about 1.8 percent of the world’s carbon dioxide. Ammonia is a colorless gas with a distinctive pungent odor that is particularly irritating to the eyes and upper respiratory system at atmospheric pressure. Because it is about half as dense as air, it rises quickly.
Indeed, ammonia does a better job of holding hydrogen than hydrogen gas itself, which is renowned for leaking through container metal walls, embrittling steel it comes into touch with, and liquefying at cryogenic temperatures with a lot of energy.

Then there’s density: all other things being equal, one and a half times more hydrogen is in a gallon of ammonia than there is in a gallon of hydrogen. As a result, ammonia is the most greenhouse gas-intensive chemical process on the planet, accounting for nearly 5% of total world CO2 emissions.
Furthermore, ammonia demand is predicted to rise in the coming years, owing to its use in synthetic fertilizers required to feed a growing worldwide population. Humans are poisoned by ammonia. In high quantities, it’s caustic, and in the United States, it’s classified as an “extremely hazardous substance,” with severe reporting requirements for any institution that uses a substantial amount.
Large-scale storage tanks carrying millions of gallons of anhydrous ammonia with a network of pipelines for off-loading to train or truck make large-scale ammonia use possible. Its use in fertilizers, explosives, as an intermediate in chemical production, metallurgy, paper/pulp manufacturing, and refrigeration exposes workers to it.
To meet net-zero standards by 2050, the quantity of carbon dioxide produced during the ammonia manufacturing process must be reduced. When generating ammonia, using low-carbon hydrogen is the greatest strategy to reduce carbon emissions. When ammonia is dissolved in water, it generates a strongly alkaline ammonium hydroxide solution, NH4OH. In many ways, the univalent radical, like K+ and Na+, reacts vigorously with acids to generate salts.
Green ammonia production could provide more possibilities in the transition to net-zero carbon dioxide emissions. These include:
- Energy storage – At low pressures (10-15 bar), ammonia can be stored in bulk as a liquid or chilled to -33°C. As a result, it’s an excellent chemical storage system for renewable energy. Ammonia is currently stored in massive chilled tanks and moved around the world by pipes, road tankers, and ships, according to an existing distribution network.
- Zero-carbon fuel – To generate power, ammonia can be burned in an engine or used in a fuel cell. The sole by-products of ammonia usage are water and nitrogen. The maritime industry is likely to be an early adopter, with fuel oil in marine engines being phased out.
- Hydrogen carrier – Hydrogen gas is employed in some applications (for example, in PEM fuel cells), but it is difficult and expensive to store in mass (needing cryogenic tanks or high-pressure cylinders). Ammonia is easier to store and carry, and it can easily be cracked and refined to produce hydrogen gas when needed.
Ammonia was produced in the nineteenth century by distilling coal tar. The value of nitrogen fertilizers in agriculture was recognized in the mid-1800s, and this, combined with the expansion of the chemical industry, provided motivation to develop a nitrogen-fixing technology. Ammonia production is currently a polluting and energy-intensive process.
The majority of hydrogen produced today comes from steam-reformed methane gas. The extraction of natural gas from the earth invariably results in methane leakage into the atmosphere, where it acts as a powerful greenhouse gas, and the steam reforming process not only consumes a lot of energy but also releases carbon dioxide. Ammonia solution is usually present in a formulation as a highly diluted form when utilized as an excipient.
Information Sources: