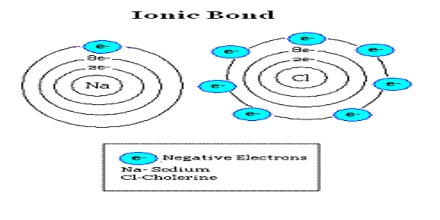
General purpose of this lecture is to describe on Ionic Bonds and Compounds. Ionic Bonds can transfer of electron (e-) from one atom to another. It is observed because metals with few electrons in its outer-most orbital. Here briefly describe on properties of Ionic Compounds: Crystalline solids at room temperature, Arranged in 3-D patterns, Large attractive forces, High melting points and Conduct electricity (dissolved in H2O).
Ionic Bonds and Compounds