Lithium aluminum hydride (LAH), is an inorganic compound with the chemical formula LiAlH4. It appears as a white powder that turns gray when exposed to air. It’s a dark grey solid. Finholt, Bond, and Schlesinger discovered it in 1947. This compound is used in organic synthesis as a reducing agent, particularly for the reduction of esters, carboxylic acids, and amides. The solid is extremely reactive to water, emitting gaseous hydrogen (H2). Some related derivatives for hydrogen storage have been discussed. Friction can cause ignition if it is spread out over a large flat combustible surface.
Although LAH is a colorless solid, commercial samples are typically gray due to contamination. Recrystallization from diethyl ether can be used to purify this material. A Soxhlet extractor is used in large-scale purifications. Because the impurities are innocuous and easily separated from the organic products, impure gray material is commonly used in synthesis. The pure powdered material, but not the large crystals, is pyrophoric. Mineral oil is used in some commercial materials to inhibit reactions with atmospheric moisture, but it is more commonly packed in moisture-proof plastic sacks.
Properties
LAH is a colorless, odorless crystalline solid with a density of 0.97 g/mL and a melting point of 150 °C. It is also commercially available as a dispersion in mineral oil to avoid its violent reaction with atmospheric moisture.
- Melting point: 125 °C (dec.)(lit.)
- Boiling point: 0°C
- Density: 0.97 g/mL at 20 °C
- Flash point: 99 °F
- Specific Gravity: 0.917
- Color: White to light gray
- Odor: Odorless solid
- Water Solubility: Reacts

Structure
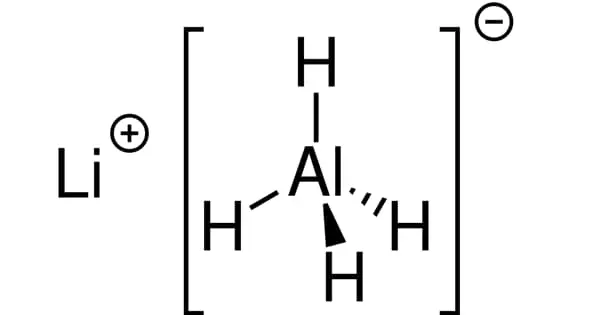
Lithium aluminum hydride has the chemical formula LiAlH4 and a molar mass of 37.95 g/mol. LAH’s chemical structure is depicted below. It is a monoclinic crystalline solid composed of lithium cation (Li+) and aluminum hydride anion (AlH4-), with Al covalently bonded to the four H atoms.
Preparation
LAH violently reacts with water, including atmospheric moisture, to liberate dihydrogen gas. The reaction proceeds according to the following idealized equation:
LiAlH4 + 4 H2O → LiOH + Al(OH)3 + 4 H2
This reaction provides a useful method to generate hydrogen in the laboratory. Aged, air-exposed samples often appear white because they have absorbed enough moisture to generate a mixture of the white compounds lithium hydroxide and aluminum hydroxide.
LiAlH4 was first prepared from the reaction between lithium hydride (LiH) and aluminum chloride:
4 LiH + AlCl3 → LiAlH4 + 3 LiCl
In addition to this method, the industrial synthesis entails the initial preparation of sodium aluminum hydride from the elements under high pressure and temperature:
Na + Al + 2 H2 → NaAlH4
LiAlH4 is then prepared by a salt metathesis reaction according to:
NaAlH4 + LiCl → LiAlH4 + NaCl
which proceeds in a high yield. LiCl is removed by filtration from an ethereal solution of LAH, with subsequent precipitation of LiAlH4 to yield a product containing around 1% w/w LiCl.
An alternative preparation starts from LiH, and metallic Al instead of AlCl3. Catalyzed by a small quantity of TiCl3 (0.2%), the reaction proceeds well-using dimethyl ether as solvent. This method avoids the cogeneration of salt.
Uses
It is primarily used as a reducing agent in organic synthesis and as a reagent in the synthesis of a wide range of pharmaceuticals, organic chemicals, and metal hydrides (such as NaH). It’s also a polymerization catalyst and a propellant. It is being investigated as a possible hydrogen storage agent in fuel cells.
Health effects/safety hazards
LAH has a strong irritant effect on the eyes, skin, mucous membranes, and respiratory system. The solid can cause severe burns to the eyes and skin. It is extremely dangerous when inhaled. Its violent reaction to water is also a hazard to one’s safety.