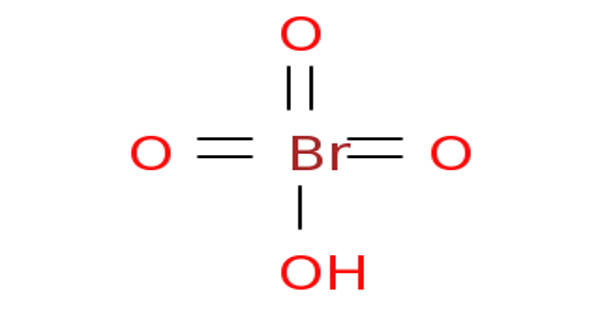
The compound perbromic acid is the inorganic compound with the formula HBrO4. It’s a strong acid and also a powerful oxidizing agent. It is an oxoacid of bromine. It is the conjugate acid of a perbromate.
Perbromic acid is an unstable compound that cannot be created by displacing chlorine from perchloric acid, as periodic acid is; it can only be created by protonation of the perbromate ion. Prior to 1968, synthesis was not possible, and it was only in the last few years that a suitable method that did not require the use of fluorine as an oxidizing agent was discovered (through the reaction of hypobromite and bromate ions in an alkaline sodium hypobromite solution, to form perbromate anions in 2011). The acid can then be formed by protonating the perbromate.
Structure
Perbromic acid’s 2D chemical structure image is also known as the skeletal formula, which is the standard notation for organic molecules. In the chemical structure of perbromic acid, the carbon atoms are implied to be located at the corner(s), and hydrogen atoms attached to carbon atoms are not indicated – each carbon atom is assumed to be associated with enough hydrogen atoms to provide the carbon atom with four bonds.
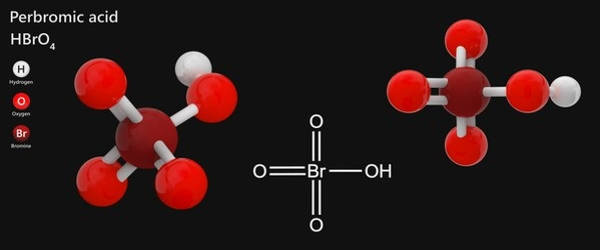
Perbromic acid’s 3D chemical structure image is based on the ball-and-stick model, which depicts both the three-dimensional position of the atoms and the bonds between them. The radius of the spheres is thus smaller than the rod lengths in order to provide a clearer view of the atoms and bonds throughout the perbromic acid chemical structure model.
Preparation
Perbromic acid is unstable and is prepared by perbromate ion protonation. Due to its instability, it cannot be synthesized by displacement reaction(by displacing chlorine of perchloric acid).
These are the chemical properties of perbromic acid:
- It is a strong oxidizing agent.
- It promptly decomposes on standing to oxygen and bromic acid (it is the most unstable among the halogen (VII) oxoacids).
- It interacts with bases to produce perbromate salts.
Perbromic acid is a strong acid and strongly oxidizing. It is the most unstable of the halogen(VII) oxoacids. It decomposes rapidly on standing to bromic acid and oxygen. So, perbromic acid is a strong acid that decomposes readily. But under the right circumstances, either it or one of its decomposition products can serve as a strong oxidizer. It reacts with bases to form perbromate salts.
Information Source: