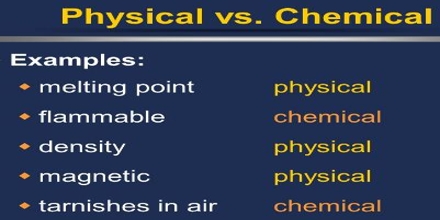
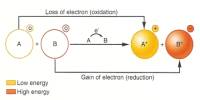
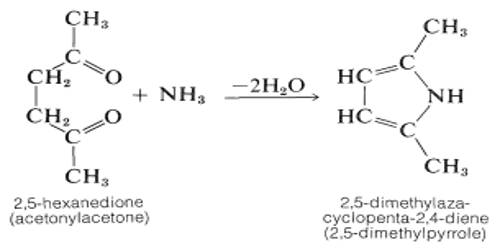
Prime purpose of this lecture is to present on Physical Properties vs Chemical Properties. Physical properties can be measure without changing the basic identity of the substance. Examples of physical properties are: color, smell, freezing point, boiling point, melting point, infra-red spectrum etc. Chemical properties describe how substances react or change to form different substances. Example: hydrogen burns in oxygen. Here also present on Physical and Chemical Changes.