Researchers created rechargeable batteries that have a six-fold increase in charge storage capacity. An international team of researchers led by Stanford University has developed rechargeable batteries that can store up to six times the charge of commercially available batteries.
The breakthrough, described in a new paper published in the journal Nature, could accelerate the use of rechargeable batteries and bring battery researchers one step closer to achieving two of their field’s top stated goals: developing a high-performance rechargeable battery that could allow cellphones to be charged only once a week rather than daily, and developing electric vehicles that can travel six times farther than gasoline vehicles.
The new alkali metal-chlorine batteries, developed by a team led by Stanford chemistry Professor Hongjie Dai and doctoral candidate Guanzhou Zhu, rely on the chemical conversion of sodium chloride (Na/Cl) or lithium chloride (Li/Cl2) to chlorine.
When electrons travel from one side of a rechargeable battery to the other, the chemistry returns to its original state, ready for another use. Non-rechargeable batteries, on the other hand, are out of luck. Their chemistry cannot be restored once they have been drained.
It is a high-performance rechargeable battery that could enable cellphones to be charged only once a week instead of daily and electric vehicles that can travel six times farther without a recharge.
“A rechargeable battery is a bit like a rocking chair. It tips in one direction, but then rocks back when you add electricity,” Dai explained. “What we have here is a high-rocking rocking chair.”
Serendipitous discovery
Because chlorine is too reactive and difficult to convert back to chloride with high efficiency, no one has yet developed a high-performance rechargeable sodium-chlorine or lithium-chlorine battery. The battery performance was poor in the few cases where others were able to achieve some degree of rechargeability.
In fact, Dai and Zhu’s goal was not to develop rechargeable sodium and lithium-chlorine battery at all, but rather to improve their existing battery technologies with thionyl chloride. This chemical is a key component of lithium-thionyl chloride batteries, a popular type of single-use battery that was first developed in the 1970s.
However, the Stanford researchers noticed that the conversion of one chemical to another had somehow stabilized, resulting in some rechargeability in one of their early experiments involving chlorine and sodium chloride. “I didn’t believe it was possible,” Dai admitted. “It took us at least a year to figure out what was going on.”

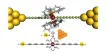
Over the next several years, the team elucidated the reversible chemistries and sought ways to improve efficiency by experimenting with a variety of positive electrode materials. The big breakthrough came when they formed the electrode with collaborators Professor Yuan-Yao Li and his student Hung-Chun Tai from Taiwan’s National Chung Cheng University using advanced porous carbon material. The carbon material has a nanosphere structure with numerous ultra-small pores. In practice, these hollow spheres act like sponges, sucking up large amounts of otherwise volatile chlorine molecules and storing them inside the micropores for later conversion to salt.
“When the battery is charged, the chlorine molecule is trapped and protected in the tiny pores of the carbon nanospheres,” Zhu explained. “When the battery needs to be drained or discharged, we can discharge it and convert chlorine to NaCl (table salt), a process that can be repeated many times. We can currently cycle up to 200 times and there is still room for improvement.”
The end result is a step closer to the battery’s brass ring—high energy density. The researchers have so far achieved 1,200 milliamp hours per gram of positive electrode material, whereas commercial lithium-ion batteries today have capacities of up to 200 milliamp hours per gram. “Ours is at least six times larger,” Zhu said.
The researchers hope to use their batteries in situations where frequent recharging is not practical or desirable, such as satellites or remote sensors. Many otherwise usable satellites are now floating in space, rendered inoperable by dead batteries. Future satellites with long-lasting rechargeable batteries could be outfitted with solar chargers, greatly extending their usefulness.
However, for the time being, the working prototype they’ve created may still be suitable for use in small everyday electronics such as hearing aids or remote controls. Much more work is needed to engineer the battery structure, increase the energy density, scale up the batteries, and increase the number of cycles for consumer electronics or electric vehicles.