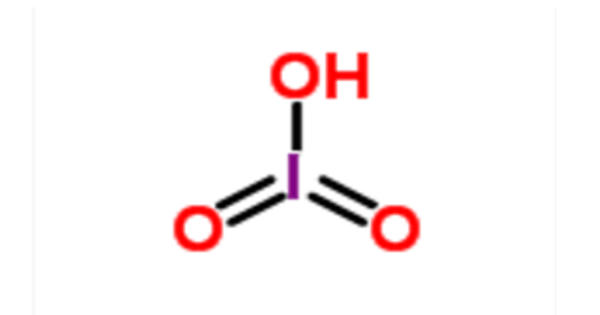
Strontium carbonate (SrCO3) belongs to the strontium carbonate salt which is found as mineral strontianite in nature. It is a powder which is dry, odorless, and tasteless. It is a weak base being a carbonate and is thus volatile with acids. Working with these is otherwise secure and safe. It is essentially insoluble in water (1 in 100,000 parts). If the water is polluted with carbon dioxide, the solubility would increase greatly, to 1 element in 1,000. Soluble in dissolved acids. It is applicable in a number of industries.
Strontium carbonate is very insoluble in water; it dissolves in acids, e.g. in hydrochloric acid, producing the following carbon dioxide:
SrCO3 + 2 HCl → SrCl2 + H2O + CO2
Strontium is in the Alkaline Earth Metals group. Such as calcium, which is in the same category, it is non-toxic. However, the chemical resemblance between strontium and calcium is due to the fact that the radioactive strontium isotopes produced during the Chernobyl reactor disaster were able to accumulate in the bones in order to cause cancer.
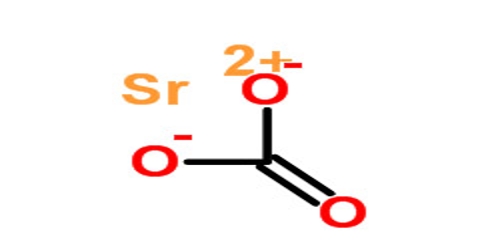
Structure of Strontium Carbonate
At present, strontium carbonates are commonly being applied as a cheap colorant in pyrotechnics since strontium and its salts produce a crimson read flame. Strontium carbonate, in general, is preferred in fireworks, compared with other strontium salts thanks to its inexpensive cost, nonhygroscopic property, and skill to neutralize the acid. It may also be used in the processing of iridescent glass, luminous paints, strontium oxide or strontium salts, and in the manufacturing of sugar and some drugs as well as road flares.
Other than the natural occurrence as a mineral, strontium carbonate is ready synthetically in one in all two processes, both of which start with present celestine, a mineral variety of strontium sulfate (SrSO4). Celesite is roasted with coke at 110–1300 ° C for forming strontium sulfide in the “black ash” phase. It’s also suggested to manufacture matte glazes as a replacement for barium. In addition, its applications include in the ceramics industry, where it serves as an ingredient in glazing, and in electrical devices, where it is used in the manufacture of strontium ferrites to produce permanent magnets for loudspeakers and door magnets.
A mixture of strontium sulfide with either carbon dioxide gas or sodium carbonate then results in the formation of a strontium carbonate precipitate.
SrS + H2O + CO2 → SrCO3 + H2S
SrS + Na2CO3 → SrCO3 + Na2S
The crystal structure is orthorhombic, with group space Pmcn and group point 2/m, 2/m, 2/m. Strontium carbonate only has one stable form (aragonite-type structure) and precipitation temperature has no effect on crystal shape, unlike calcium or magnesium carbonate.
Among other things, strontium carbonate is used to produce ferrite magnets which are used to remove strontium ferrite. Its main use is the processing of glass for cathode-ray tubes, which are best known as television tubes (color). Since strontium carbonate has a relatively broad atomic radius, the X-radiation found in the tubes is absorbed.
Strontium carbonate is like nanometer-sized powder, not self-inflammable. It is also not inflammable as a mixture of air (dust) under the control of an ignition source so there is no risk of explosion of dust. Strontium carbonate is also used for the production of some superconductors such as BSCCO and also for products with electroluminescent properties.
Strontium is called after the mineral strontianite which, in turn, is called after the situation of Strontian, Scotland, where the primary strontium mineral was discovered. Due to its status as a weak Lewis base, strontium carbonate are often wont to produce many alternative strontium compounds by the simple use of the corresponding acid. At 1,200° C (2,192° F) strontium carbonate decomposes to form strontium oxide which CO2, and is dissolved by excess CO2, producing Sr(HCO3)2 strontium bicarbonate solution. The strontium occurs inside the host calcite as strontianite in a solid solution with a strontium content of up to one percent.
Information Sources: