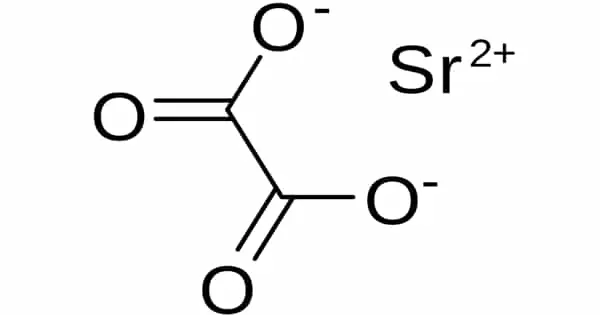
Strontium oxalate is a chemical compound with the formula SrC2O4. It is a fine white, insoluble powder that forms as a result of precipitation. The high water content could be a disadvantage. It is occasionally used to obtain red colors (though it is more expensive than carbonate) and in some glitter formulas with gunpowder and Al.
Strontium oxalate can exist as a hydrated compound (SrC2O4•nH2O) or as an acidic salt (SrC2O4•mH2C2O4•nH2O). It should not be used in chlorate mixtures and should be kept away from acids.
Properties
Strontium oxalate is a white precipitate formed by the reaction of strontium salt solution and ammonium oxalate solution. It crystallizes from hot water on cooling. The solubility at ~25o in H2O is 1g/20L, and in 3.5% AcOH and 25% AcOH it is 1g/1.9L and 1g/1.1L respectively.
- Compound Formula: SrC2O4
- Molecular Weight: 175.65
- Appearance: White Powder
- Color: white
- Melting Point: N/A
- Boiling Point: N/A
- Density: N/A
- Solubility in H2O: N/A
- Exact Mass: 175.885 g/mol
- Solubility: Soluble in hydrochloric acid and nitric acid

Preparation
Strontium oxalate exists in two different forms: the neutral strontium oxalate hydrate, SrC2O4·xH2O, and the acid salt of strontium oxalate, SrC2O4·yH2C2O4·xH2O. Depending on the concentration of oxalic acid and ammonium oxalate as precipitating agents, both forms can be obtained. It should be avoided in chlorate mixtures and kept away from acids.
Use in pyrotechnics
With the addition of heat, strontium oxalate will decompose based on the following reaction:
SrC2O4 → SrO + CO2 + CO
Strontium oxalate is an excellent pyrotechnic agent because it decomposes quickly when heated. It produces a red color when it decomposes into strontium oxide. Strontium oxalate is an excellent red color-producing agent in the presence of magnesium because this reaction produces carbon monoxide, which can be further reduced with magnesium oxide. If magnesium is not present, strontium carbonate has been found to be a better option for producing an even greater effect.
Hazards: Strontium oxalate is harmful by skin contact, inhalation, and ingestion as it is a salt of oxalic (ethanedioic) acid, it can cause renal failure and may even prove to be fatal if ingested. It should not be used in chlorate mixtures and should be kept away from acids.