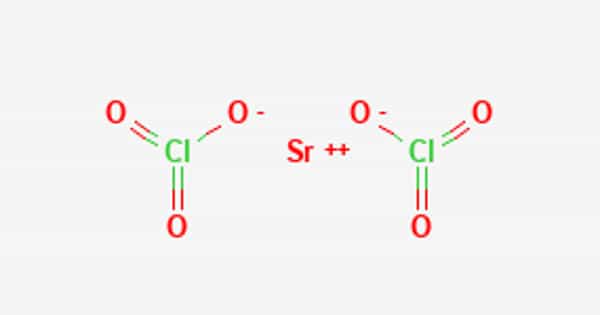
Strontium perchlorate, also known as Sr(ClO4)2, is a white powder or colorless crystal. It has the appearance of a colorless crystalline powder. It is hygroscopic and has been used in sealed pyrotechnic devices to produce a red color. Electrolysis can be used to create it. When in contact with organic materials, it poses a serious fire risk. It is used in the production of other chemicals.
It is a strong oxidizer that gives red flames. It can be used in pyrotechnics; however, usually the more common strontium nitrate is used. It is also used in Liquid Injection Thrust Vector Control (LITVC) in solid-propellant rockets to enable steering control with a simply fixed nozzle.
Properties
- Compound Formula: Cl2O8Sr
- Molecular Weight: 288.52
- Appearance: White crystalline powder
- Melting Point: < 100 °C
- Boiling Point: N/A
- Density: N/A
- Solubility in H2O: N/A
- Exact Mass: 285.802634 g/mol

Preparation
It can be prepared by oxidizing strontium chlorate with hypochlorites.
Strontium perchlorate has the formula, Sr(ClO4)2 and a molecular weight of 286.52 g/mol. It is a white solid with a melting point of 100°C. Its density is 2.073 g/cm3 and it is slightly soluble in water (3.21 g/100 ml at 20°C). It can be prepared by reaction of strontium carbonate with a solution of perchloric acid or ammonium perchlorate:
SrCO3 + 2HClO4 → Sr(ClO4)2 +H2O+ CO2
The hexahydrate is formed in an aqueous solution. It is a deliquescent crystalline mass that is soluble in water and alcohol. The alcohol solution emits a crimson flame when ignited. When strontium chlorate is heated slightly above 290° C, the salt is also formed.
Reactivity Profile
It can react with reducing agents to produce heat and possibly gaseous products (causing pressurization of closed containers). The products may be capable of further reactions (such as combustion in the air). Chemical reduction of materials in this class can be rapid or even explosive, but it frequently necessitates initiation (heat, spark, catalyst, addition of a solvent).
If initiation is prevented, explosive mixtures of inorganic oxidizing agents and reducing agents can often remain unchanged for long periods of time. Such systems are typically solid mixtures, but they can include any combination of physical states. Some inorganic oxidizing agents are metal salts that are soluble in water; dissolution dilutes but does not eliminate such materials’ oxidizing power.
Organic compounds, in general, have some reducing power and can, in theory, react with other organic compounds. The actual reactivity of an organic compound varies greatly depending on its identity. Active metals, cyanides, esters, and thiocyanates can all react violently with inorganic oxidizing agents.
Health Hazard
Inhaling, ingesting, or coming into contact (skin, eyes) with vapors or substances can result in severe injury, burns, or death. Fire can emit irritant, corrosive, and/or toxic gases. Pollution may occur as a result of runoff from firefighting or dilution of water.
Fire Hazard
When these substances are involved in a fire, they will accelerate the burning process. When heated or involved in a fire, some may decompose explosively. It may explode as a result of heat or contamination. Some will explode when combined with hydrocarbons (fuels). It may cause combustibles to ignite (wood, paper, oil, clothing, etc.). When heated, containers may explode. Runoff can cause a fire or explosion.