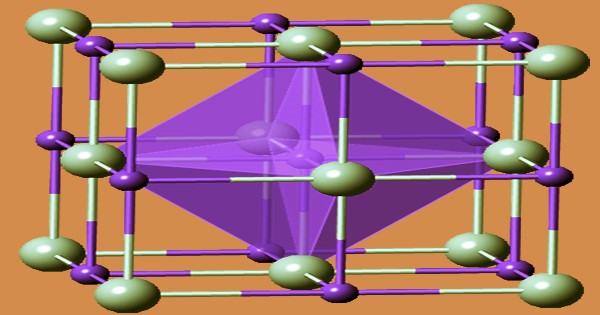
Tantalum carbide (TaC) is made principally by reducing Ta2O5 with carbon at temperatures ranging from 1600 to 2000 degrees Celsius. It belongs to a group of tantalum and carbon binary chemical compounds with the empirical formula TaCx, where x is generally between 0.4 and 1. TaC is a gold-colored powder made by reacting carbon directly with tantalum scrap or tantalum pentoxide at temperatures up to 1,900 degrees Celsius (3,452 degrees Fahrenheit). They are refractory ceramic materials that are exceedingly hard, brittle, and have metallic electrical conductivity. They appear as brown-gray powders that are usually sintering treated.
Tantalum carbide powder is made using a technology similar to that used to make tungsten carbide powder. Tantalum carbides are major cermet materials that are commercially employed in cutting tool bits and are sometimes added to tungsten carbide alloys. The cutting edge of machine tools is made of a blend of tantalum carbide (TaC) and graphite, which is an extremely hard substance. Tantalum carbides have a melting point of around 3880 °C, which is among the highest for binary compounds, depending on purity and measurement conditions.
To minimize grain formation, TaC is added to tungsten carbide-cobalt-based cutting tools in tiny amounts (0.2–2 wt%) in the form of TaC or mixed carbides such TaNbC and WTiTaC. Only tantalum hafnium carbide has a slightly higher melting point of roughly 3942 °C, whereas hafnium carbide’s melting point is comparable to TaC’s. Before the final carbon matrix insertion, TaC was infiltrated as carbon fiber coatings. Similarly, HfC-containing multilayer coatings were infiltrated into Cf/C composites to provide an exterior coating that was deeply embedded in the material.
Heating a mixture of tantalum and graphite powders in a vacuum or inert-gas atmosphere produces TaCx particles with the desired composition (argon). A furnace or an arc-melting setup is used to heat the material to a temperature of around 2000 °C. The addition of 2–15 wt% TaC to cemented carbides improves their thermal shock resistance as well as their resistance to cratering and oxidation. Tantalum pentoxide can also be reduced by carbon in a vacuum or hydrogen environment at temperatures between 1500 and 1700 degrees Celsius. Several subcarbides can be seen in the tantalum–carbon phase diagram.
Self-propagating high-temperature synthesis has been used to produce TaC directly from the elements. When it comes to metal cutting, there are two significant phenomena. One is that the temperature at the cutting tip, which is the point where the cutting tool meets the workpiece, can reach 1000°C or even higher. Tantalum carbides have a complex mixture of ionic, metallic, and covalent interaction between tantalum and carbon atoms, and these carbides are exceedingly hard and brittle due to the strong covalent component.
Tantalum carbide and tungsten carbide in a nickel, cobalt, and/or chromium matrix create a RHM formulation that is particularly well suited for corrosion and wear resistance. In terms of magnitude and temperature dependency, tantalum carbides have metallic electrical conductivity. TaC is a superconductor with a comparatively high TC = 10.35 K transition temperature. TaCx has diamagnetic magnetic characteristics for x ≤ 0.9 and paramagnetic magnetic characteristics for x. Despite having the same crystal structure as TaCx, HfCx exhibits an inverted behavior (para-diamagnetic transition with increasing x).
Nozzles, orifice plates, and valve components are examples of applications where some grades are almost as corrosion resistant as platinum. Due to its excellent physical properties in melting point, hardness, elastic modulus, thermal conductivity, thermal shock resistance, and chemical stability, tantalum carbide is widely used as a sintering additive in ultra-high temperature ceramics (UHTCs) or as a ceramic reinforcement in high-entropy alloys (HEAs). This makes it a desirable material for aircraft and rockets in the aerospace industry.
Information Sources: