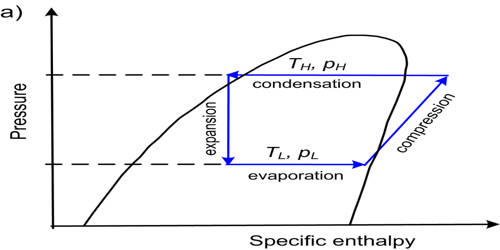
A transcritical cycle is a thermodynamic cycle where the working fluid goes through both Subcritical and supercritical states. It is a cycle with a maximum pressure higher than the critical one. This is often the case when carbon dioxide, CO2, is the refrigerant. In transcritical systems, CO2 is cooled but does not condense at the gas cooler outlet, being above the critical temperature. The use of carbon dioxide as operating fluid in vapor compression refrigeration systems is nowadays receiving new attention, due to its environmental impact but also due to unexpectedly good performance. Transcritical CO2 cycle is recently considered as one of the most influential refrigerants for its characteristics such as non-ODP, negligible GWP, non-flammability and non-toxicity, despite the drawback of high working pressure. The phase change is gradual and all the physical and thermodynamic properties vary without discontinuity in the heat introduction process.
Advantages:
- the system that completely uses natural refrigerants (CO2);
- various studies have shown higher efficiency than any other type of installation at average outside temperatures less than 15°C;
- technology becoming standardized, costs are falling.
Critical:
- high pressures involved (up to 120 barg);
- systems are normally more complex than traditional ones;
- good efficiency even in warm climates, using additional technology such as parallel compressors.
The modern transcritical cycle was developed in 1988–1991 by the Norwegian professor Gustav Lorentzen (1915–1995) and his team. CO2 is a component of our atmosphere that is essential to life. It has no ozone depletion potential and insignificant global warming potential, so CO2 has no regulatory liability, as do HFCs. A transcritical cycle differs from a conventional phase-change vapor compression cycle as for the heat rejection phase in the high-temperature cycle section, which is not operated through a condenser, being replaced by a gas cooler, where the CO2 temperature decreases over a large temperature range. They are interesting because they can achieve higher efficiency compared to the subcritical ones if a finite heat capacity source is available.