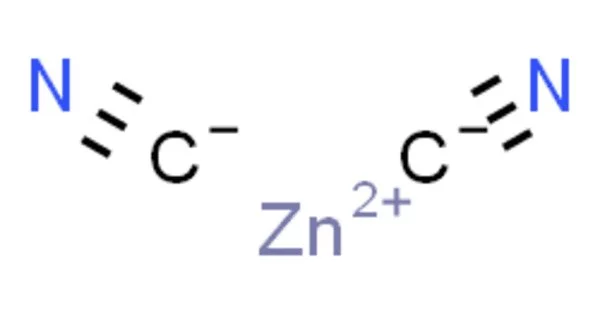
The inorganic compound zinc cyanide has the formula Zn(CN)2. It has the appearance of a white powder. It is a white solid that is primarily used for electroplating zinc but also has more specialized applications in organic compound synthesis. It is not water soluble. Inhalation (dust and hydrogen cyanide from slight decomposition) and ingestion are both toxic.
Zinc cyanide is used in electroplating, insecticides, and ammonia separation from producer gas. It is used as an electroplating reagent and as a reagent. It can also be found in medicine, metal plating, and chemical analysis.
Properties
White powder; orthorhombic crystals; density 1.852 g/cm3; decomposes at 800°C; insoluble in water (about 5mg/L at 20°C); soluble in alkalies, potassium cyanide and ammonia solutions; insoluble in alcohol.
- Chemical formula: Zn(CN)2
- Molar mass: 117.444 g/mol
- Appearance: white solid
- Density: 1.852 g/cm3, solid
- Melting point: 800 °C (1,470 °F; 1,070 K) (decomposes)
- Solubility in water: 0.0005 g/100 mL (20 °C)
- Solubility: attacked by alkalies, KCN, ammonia

Chemical properties
Typical for an inorganic polymer, Zn(CN)2 is insoluble in most solvents. The solid dissolves in, or more precisely, is degraded by, aqueous solutions of basic ligands such as hydroxide, ammonia, and additional cyanide to give anionic complexes.
Structure
Zinc adopts a tetrahedral coordination environment in Zn(CN2)2, which is linked by bridging cyanide ligands. Two “interpenetrating” structures make up the structure (blue and red in the picture above). These motifs are sometimes referred to as “expanded diamondoid” structures. Some forms of SiO2 have a similar structure, with the tetrahedral Si centers connected by oxides. The cyanide group exhibits head to tail disorder, with each zinc atom having one to four carbon neighbors and the remaining being nitrogen atoms. It has one of the highest negative thermal expansion coefficients (exceeding the previous record holder, zirconium tungstate).
Synthesis
Zn(CN)2 is easy to make by combining aqueous solutions of cyanide and zinc ions, for example via the double replacement reaction between KCN and ZnSO4:
ZnSO4 + 2 KCN → Zn(CN)2 + K2SO4
For commercial applications, some effort is made to avoid halide impurities by using acetate salts of zinc:
Zn(CH3COO)2 + HCN → Zn(CN)2 + 2 CH3COOH
Zinc cyanide is also produced as a byproduct of certain gold extraction methods. Procedures to isolate gold from aqueous gold cyanide sometimes call for the addition of zinc:
2 [Au(CN)2]– + Zn → 2 Au + Zn(CN)2 + 2 CN–
Applications
- Electroplating
The main application of Zn(CN)2 is for electroplating of zinc from aqueous solutions containing additional cyanide.
- Organic synthesis
Zn(CN)2 is used in the Gatterman reaction to introduce the formyl group into aromatic compounds, where it is a more convenient, safer, and non-gaseous alternative to HCN. Because the reaction uses HCl, Zn(CN)2 also provides the reaction with ZnCl2, a Lewis acid catalyst, in situ. Zn(CN)2 has been used in the synthesis of 2-hydroxy-1-naphthaldehyde and mesitaldehyde, for example.
Zn(CN)2 is also used as a cyanosilylation catalyst for aldehydes and ketones.