The ferrierite group of zeolite minerals (the FER structure) consists of three very similar species: ferrierite-Mg, ferrierite-Na, and ferrierite-K, based on the dominant cation in the A location. ferrierite-Mg and ferrierite-K are orthorhombic minerals and ferrierite-Na is monoclinic with highly variable cationic composition, (Na, K)2Mg(Si, Al)18O36(OH)·9H2O. Calcium and other ions are often also present. They are found in vitreous to pearly, often radiating, spherical aggregates of thin blade-shaped transparent to translucent crystals.
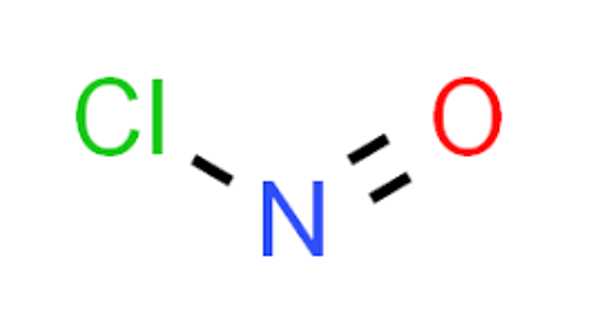
Ferrierite-H can be used as a catalyst in the chemical industry for the acid-catalyzed skeletal isomerization of n-butenes to isobutene, the raw material for the production of methyl tert-butyl ether (MTBE).
General Information
- Category: Zeolite
- Formula: (“A” position)3-5Mg[Al5-7Si5-31O72]·18H2O
- Crystal system: Orthorhombic
- Crystal class: Dipyramidal (mmm)

Fig: Ferrierite group of zeolite minerals
Properties
- Color: white, colorless, pinkish, orange to red
- Mohs scale hardness: 3 – 3.5
- Streak: white
Occurrence
Ferrierite typically occurs as an alteration mineral in basaltic rocks and in tuffaceous sediments. In North America, it is found at Kamloops Lake, BC, Canada (the original type locality) and Leavitt Lake, California. Ferrierite was named after Canadian geologist and mining engineer Walter Frederick Ferrier (1865–1950).
Information Source: