The major goal shared by the fields of virology and pharmaceutics is the development of highly effective, broad-spectrum antiviral agents. The development of antiviral drugs has focused on preventing viral entry and replication as well as modulating the cellular defense system. High throughput molecule screening, peptide genetic engineering, and functional agent screening have identified promising candidates for the development of optimal broad-spectrum antiviral agents to intervene in viral infection and control viral epidemics.
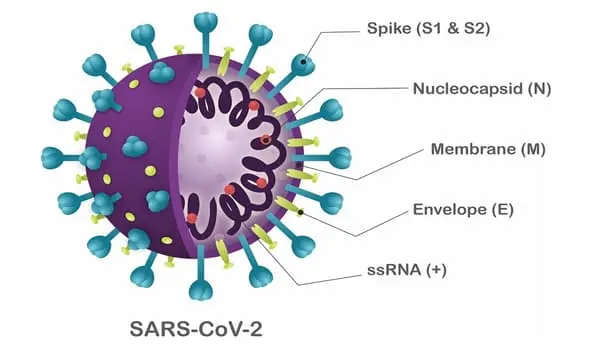
In a new study, researchers report that 4′-fluorouridine (4′-FlU), a new candidate ribonucleoside analog, has potent antiviral activity against SARS-CoV-2, RSV, and other respiratory RNA viruses in cell culture, human organoids, and different animal models when administered orally once daily.
The COVID-19 pandemic, as well as the resurgence of infections caused by other respiratory RNA viruses such as a respiratory syncytial virus (RSV), in children, has created an urgent need for the development of orally available broad-spectrum antiviral therapeutics.
The global COVID-19 outbreak poses a significant risk to humans. Antiviral therapeutics against SARS-CoV2 are desperately needed. Screening existing broad-spectrum antiviral agents for activity against SARS-CoV2 is one strategy for discovering effective antiviral drugs. In addition to COVID-19, many other epidemic viral diseases, such as influenza, dengue fever, Zika and Ebola virus infections, and western/eastern equine encephalitis, are still incurable.
Mechanistically, we show that 4′-FlU is in a different class than molnupiravir, which is currently under consideration for regulatory approval. 4′-FlU does not act as a mutagen, but instead causes the viral polymerase to terminate, preventing viral genome replication. There is an urgent need to expand the therapeutic arsenal against SARS-CoV-2, and 4′-FlU as a companion drug has strong developmental promise.
Dr. Richard Plemper
Researchers from Georgia State University’s Institute for Biomedical Sciences report a new candidate ribonucleoside analog, 4′-fluorouridine (4′-FlU), that has potent antiviral activity against SARS-CoV-2, RSV, and other respiratory RNA viruses in cell culture, human organoids, and different animal models when administered orally once daily.
A priority is the development of broad-spectrum antiviral agents to treat epidemic diseases caused by emerging and re-emerging viruses. The main approaches to this end are based on targeting viral infectivity and modulating host-defense systems. The antiviral mechanisms of broad-spectrum antiviral candidates should be thoroughly investigated in the context of various viral diseases. This Special Issue aims to collect research on broad-spectrum antiviral agents, including repositioning of existing agents, herbal components, synthesized compounds, and genetically engineered molecules.
“Mechanistically, we show that 4′-FlU is in a different class than molnupiravir, which is currently under consideration for regulatory approval,” said Dr. Richard Plemper, senior author of the study, Distinguished University Professor, and director of Georgia State’s Center for Translational Antiviral Research. “4′-FlU does not act as a mutagen, but instead causes the viral polymerase to terminate, preventing viral genome replication. There is an urgent need to expand the therapeutic arsenal against SARS-CoV-2, and 4′-FlU as a companion drug has strong developmental promise.”
4′-FlU was tested in the study against various SARS-CoV-2 variants of concern in ferrets, which have emerged as a leading model for drug testing, and against the respiratory syncytial virus in mice. The researchers discovered that this drug effectively inhibited SARS-CoV-2 replication in ferrets, including the gamma and delta variants, and effectively suppressed RSV burden in mouse lungs.
“We are excited that 4′-FlU is the only orally available antiviral candidate currently developed against SARS-CoV-2 that is active when given once daily,” said Dr. Julien Sourimant, first author of the study and a researcher in Dr. Plemper’s lab at the Institute for Biomedical Sciences, “which should be a major asset in ensuring outpatient compliance.”
The Georgia State University team collaborated with researchers from Emory University and the Texas Biomedical Research Institute on this 4′-FlU research. The study was supported by public health service grants from the National Institutes of Health/National Institute of Allergy and Infectious Diseases to Georgia State University.
Severe acute respiratory syndrome (SARS), Middle East respiratory syndrome (MERS), Western/Eastern equine encephalitis (WEE/EEE), and Ebola, as well as pandemic influenza A (H1N1), were all epidemic viral diseases that occurred in 2003, 2012, 2013, 2014, and 2009, respectively. Emerging SARS, H1N1, and MERS, as well as re-emerging WEE/EEE and EBOVes, are all lethal and spreadable via travelers. The United States has designated them as high-priority biodefense pathogens. The high virulence of these viruses, combined with the lack of effective treatments, has posed an ongoing threat to public health.