Hair loss in women with breast cancer who got scalp cooling before, during, and after chemotherapy is the subject of two studies published in the February 14 edition of JAMA.
Chemotherapy can cause hair loss (alopecia), which women consider to be one of the most unpleasant side effects. Scalp chilling is thought to diminish blood flow to hair follicles and chemotherapeutic drug absorption.
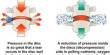
Devices that circulate fluid in a cooling cap utilizing refrigeration are used in modern procedures to prevent hair loss. Prior to chemotherapy, the patient is fitted with a cap that does not need to be replaced or removed until the treatment is over.
Although scalp cooling devices have been used to prevent alopecia, their effectiveness has yet to be shown in a randomized scientific research. Chilly caps and scalp cooling devices are tight-fitting, helmet-like headgear that you wear during chemotherapy infusions and are filled with a cold gel or liquid. Many people have been able to preserve part or all of their hair while undergoing chemotherapy, which can cause hair loss.
Julie Nangia, M.D., and colleagues from Baylor College of Medicine in Houston randomly allocated 182 women with breast cancer who were having chemotherapy to scalp cooling (n = 119) or control (n = 63) in one research.
30 minutes before, during, and 90 minutes after each chemotherapy injection, the scalp was cooled. At the completion of four rounds of chemotherapy, hair preservation was examined. A preliminary analysis was prepared to allow the research to be stopped early due to effectiveness concerns.

Cold caps and scalp cooling devices lower the quantity of chemotherapy drugs that reaches the hair follicles by constricting the blood vessels beneath the scalp’s epidermis. The hair may be less prone to fall out if there is less chemotherapy drugs in the follicles. The cold also reduces the metabolic activity of hair follicles, slowing cell division and protecting the follicles from chemotherapy.
There were 142 individuals evaluable at the time of the interim analysis. The researchers discovered that individuals who got scalp cooling had a considerably higher chance of having less than 50% hair loss than those who did not get scalp cooling (with 51 percent of those in the scalp cooling group retaining their hair, compared with 0 percent of those in the control group).
There were no significant differences between the groups in terms of changes in any of the quality of life variables. Only device-related adverse events were gathered; in the cooling group, 54 adverse events were reported, none of which were significant.
“Further research is needed to assess longer-term efficacy and adverse effects,” the authors write.
Hope S. Rugo, M.D., and colleagues from the University of California, San Francisco, included women with breast cancer who were taking chemotherapy in another trial (106 patients in the scalp cooling group and 16 in the control group; 14 matched by both age and chemotherapy regimen).
Scalp cooling was started 30 minutes before each chemotherapy cycle, and the temperature was kept at 3°C (37°F) throughout the treatment and for 90 to 120 minutes following.
Although scalp cooling has been available in Europe for decades, its use in the United States has been limited due to a number of variables, including a lack of prospective effectiveness evidence with current chemotherapy regimens and a lack of FDA certification.
Chemotherapy took an average of 2.3 months for the 122 individuals in the research. In the scalp cooling group, 67 of 101 patients (66 percent) evaluable for alopecia experienced hair loss of 50% or less, compared to 0 of 16 patients (0 percent) in the control group.
Three of the five quality-of-life indicators in the scalp cooling group were substantially improved one month following the conclusion of chemotherapy. In the scalp cooling group, 27 percent of patients said they felt less physically appealing, compared to 56 percent in the control group.
Four (3.8 percent) of the 106 patients in the scalp cooling group had a mild headache, and three (2.8 percent) stopped using the device because they were cold.
“Further research is needed to assess outcomes after patients receive anthracycline (a class of drugs used in chemotherapy) regimens, longer-term measures of alopecia, and adverse effects,” the authors write.
Cold caps and scalp cooling devices worked better for women who were treated with taxane chemotherapy alone without an anthracycline chemotherapy drug than for women who were treated with anthracycline chemotherapy. Furthermore, it implies that patients and operators have different perspectives on hair loss.