2.15 Methods used to promote improved hand decontamination
Hand-hygiene promotion has been challenging for >150 years. In-service education, information leaflets, workshops and lectures, automated dispensers, and performance feedback on hand-hygiene adherence rates have been associated with transient improvement 102.
Several strategies for promotion of hand hygiene in hospitals have been published. These strategies require education, motivation, or system change. Certain strategies are based on epidemiologic evidence, others on the authors’ and other investigators’ experience and review of current knowledge. Some strategies may be unnecessary in certain circumstances, but may be helpful in others. In particular, changing the hand-hygiene agent could be beneficial in institutions or hospital wards with a high workload and a high demand for hand hygiene when alcohol-based hand rubs are not available 18,37. However, a change in the recommended hand-hygiene agent could be deleterious if introduced during winter, at a time of higher hand-skin irritability, and if not accompanied by the provision of skin-care products (e.g., protective creams and lotions). Additional specific elements should be considered for inclusion in educational and motivational programs (Table 1).
Table 1. Methods Used To Promote Improved Hand decontamination
| Ser | Strategy | Tool for change |
| 1 | Education | Motivation and system changes |
| 2 | Routine observation and feedback | Education and motivation |
| 3 | Engineering control
| System changes |
| 4 | Patient education | Motivation and system changes |
| 5 | Reminders in the work place | System changes |
| 6 | Administrative sanction/ rewarding | System changes |
| 7 | Changes in the hand decontamination agents | System changes and eductaion |
| 8 | Promote/ facilitate skin care for HCWs hands | System changes and eductaion |
| 9 | Obtain active participation in individual and institutional level | Eductaion motivation and system changes |
| 10 | Improve institutional safety climate | Motivation and system changes |
| 11 | Enhance individual and institutional self efficacy | Eductaion motivation and system changes |
| Avoid overcrowding, understaffing and excessive overload | System changes | |
| Combine several above startegies | Eductaion motivation and system changes |
Several strategies that could potentially be associated with successful promotion of hand hygiene require a system change. Hand-hygiene adherence and promotion involve factors at both the individual and system level. Enhancing individual and institutional attitudes regarding the feasibility of making changes (self-efficacy), obtaining active participation of personnel at both levels, and promoting an institutional safety climate represent challenges that exceed the current perception of the role of infection-control professionals.
2.15.2 Elements of HCWs educational and motivational programs
2.15.2.1 Rationale for hand decontamination
- Potential risks of transmission of microorganism to patients
- Potential risks of HCWs colonization or infection caused by organisms acquired from the patients
- Morbidity, mortality and costs associated with health care associated infections.
2.15.2.2 Indication for hand decontamination
- Contact with patients intact skin( taking blood pressure, performing physical examination, lifting the bed etc)
- Contact with environmental surfaces in the immediate vicinity of patients
- After glove removal
2.15.2.3 Techniques for hand decontamination
- Amount of hand decontamination solution
- Duration of hand decontamination procedure
- Selection of hand decontamination agents
ü Alcohol based hand rubs are the most efficacious agents for reducing the number of bacteria on the hands of personnel. Antiseptic soaps are recommended and detergents are the next most effective, and non antibacterial soaps are the least effective.
ü Soap and water are recommended for visibly soil hands.
ü Alcohol based hand rubs are recommended for routine decontamination of hands for all clinical indications( except when hands are visibly soiled) and as one of the options for surgical hand decontamination.
2.15.2.4 Methods to maintain hand skin health
- Lotions and creams can prevent or minimize skin dryness and irritation caused by irritant contact dermatitis
- Acceptable lotions or creams to use
- Recommended schedule for applying lotions or creams
2.15.2.4 Expectations of patient care managers/ administrators
- Written statements regarding the value of, and support for adherence to recommended hand decontamination practices
- Role models demonstrating adherence to recommended hand decontamination practices
2.15.2.5 Indications for, and limitations of, glove use
- Hand contamination may occur as a result of small, undetected holes in gloves
- Contamination may occur during glove removal
- Wearing gloves does not replace the need for hand decontamination
- Failure to remove gloves after caring for a patient may lead to transmission of microorganism from one point to another.
Whether increased education, individual reinforcement technique, appropriate rewarding, administrative sanction, enhanced self-participation, active involvement of a larger number of organizational leaders, enhanced perception of health threat, self-efficacy, and perceived social pressure 19,105, or combinations of these factors can improve HCWs’ adherence with hand hygiene needs further investigation. Ultimately, adherence to recommended hand-hygiene practices should become part of a culture of patient safety where a set of interdependent quality elements interact to achieve a shared objective 109.
On the basis of both these hypothetical considerations and successful, actual experiences in certain institutions, strategies to improve adherence to hand-hygiene practices should be both multimodal and multidisciplinary. However, strategies must be further researched before they are implemented.
2.16 Other policies related to hand hygiene
2.16.1 Fingernails and artificial nails
Studies have documented that subungual areas of the hand harbor high concentrations of bacteria, most frequently coagulase-negative staphylococci, gram-negative rods (including Pseudomonas spp.), Corynebacteria, and yeasts 110. Freshly applied nail polish does not increase the number of bacteria recovered from periungual skin, but chipped nail polish may support the growth of larger numbers of organisms on fingernails. Even after careful handwashing or the use of surgical scrubs, personnel often harbor substantial numbers of potential pathogens in the subungual spaces 110.
An outbreak of P. aeruginosa in a neonatal intensive care unit was attributed to two nurses (one with long natural nails and one with long artificial nails) who carried the implicated strains of Pseudomonas spp. on their hands 111.
2.16.2 Gloving policies
CDC has recommended that HCWs wear gloves to 1) reduce the risk of personnel acquiring infections from patients, 2) prevent health-care worker flora from being transmitted to patients, and 3) reduce transient contamination of the hands of personnel by flora that can be transmitted from one patient to another. Before the emergence of the acquired immunodeficiency syndrome (AIDS) epidemic, gloves were worn primarily by personnel caring for patients colonized or infected with certain pathogens or by personnel exposed to patients with a high risk of hepatitis B. Since 1987, a dramatic increase in glove use has occurred in an effort to prevent transmission of HIV and other bloodborne pathogens from patients to HCWs 112. Gloves be worn during all patient-care activities that may involve exposure to blood or body fluids that may be contaminated with blood 112.
The effectiveness of gloves in preventing contamination of HCWs’ hands has been confirmed in several clinical studies 24,27. One study found that HCWs who wore gloves during patient contact contaminated their hands with an average of only 3 CFUs per minute of patient care, compared with 16 CFUs per minute for those not wearing gloves 24. Two other studies, involving personnel caring for patients with C. difficile or VRE, revealed that wearing gloves prevented hand contamination among the majority of personnel having direct contact with patients 27. Wearing gloves also prevented personnel from acquiring VRE on their hands when touching contaminated environmental surfaces. Preventing heavy contamination of the hands is considered important, because handwashing or hand antisepsis may not remove all potential pathogens when hands are heavily contaminated 49.
Several studies provide evidence that wearing gloves can help reduce transmission of pathogens in health-care settings. In a prospective controlled trial that required personnel to routinely wear vinyl gloves when handling any body substances, the incidence of C. difficile diarrhea among patients decreased from 7.7 cases/1,000 patient discharges before the intervention to 1.5 cases/1,000 discharges during the intervention 79. The prevalence of asymptomatic C. difficile carriage also decreased substantially on “glove” wards, but not on control wards. In intensive-care units where VRE or MRSA have been epidemic, requiring all HCWs to wear gloves to care for all patients in the unit (i.e., universal glove use) likely has helped control outbreaks 113.
The influence of glove use on the hand-hygiene habits of personnel is not clear. Several studies found that personnel who wore gloves were less likely to wash their hands upon leaving a patient’s room. In contrast, two other studies found that personnel who wore gloves were substantially more likely to wash their hands after patient care 107.
The following caveats regarding use of gloves by HCWs must be considered. Personnel should be informed that gloves do not provide complete protection against hand contamination. Bacterial flora colonizing patients may be recovered from the hands of <30% of HCWs who wear gloves during patient contact 27. Further, wearing gloves does not provide complete protection against acquisition of infections caused by hepatitis B virus and herpes simplex virus 114. In such instances, pathogens presumably gain access to the caregiver’s hands via small defects in gloves or by contamination of the hands during glove removal 108.
Gloves used by HCWs are usually made of natural rubber latex and synthetic nonlatex materials (e.g., vinyl, nitrile, and neoprene [polymers and copolymers of chloroprene]). Because of the increasing prevalence of latex sensitivity among HCWs and patients, FDA has approved several powdered and powder-free latex gloves with reduced protein contents, as well as synthetic gloves that can be made available by health-care institutions for use by latex-sensitive employees. In published studies, the barrier integrity of gloves varies on the basis of type and quality of glove material, intensity of use, length of time used, manufacturer, whether gloves were tested before or after use, and method used to detect glove leaks. In published studies, vinyl gloves have had defects more frequently than latex gloves, the difference in defect frequency being greatest after use. However, intact vinyl gloves provide protection comparable to that of latex gloves. Limited studies indicate that nitrile gloves have leakage rates that approximate those of latex gloves115. Having more than one type of glove available is desirable, because it allows personnel to select the type that best suits their patient-care activities. Use of petroleum-based hand lotions or creams may adversely affect the integrity of latex gloves 116. After use of powdered gloves, certain alcohol hand rubs may interact with residual powder on the hands of personnel, resulting in a gritty feeling on the hands. In facilities where powdered gloves are commonly used, various alcohol-based hand rubs should be tested after removal of powdered gloves to avoid selecting a product that causes this undesirable reaction. Personnel should be reminded that failure to remove gloves between patients may contribute to transmission of organisms .
2.16.3 Jewelry
Several studies have demonstrated that skin underneath rings is more heavily colonized than comparable areas of skin on fingers without rings 118. One study found that 40% of nurses harbored gram-negative bacilli (e.g., E. cloacae, Klebsiella, and Acinetobacter) on skin under rings and that certain nurses carried the same organism under their rings for several months. In a more recent study involving >60 intensive care unit nurses, multivariable analysis revealed that rings were the only substantial risk factor for carriage of gram-negative bacilli and S. aureus and that the concentration of organisms recovered correlated with the number of rings worn. Whether the wearing of rings results in greater transmission of pathogens is unknown. Two studies determined that mean bacterial colony counts on hands after handwashing were similar among persons wearing rings and those not wearing rings. Further studies are needed to establish if wearing rings results in greater transmission of pathogens in health-care settings118.
Methods and Materials
To determine compliance of hand decontamination practices the study was carried out as per following methodology:
Here, n= Desired sample size
z= Standard normal deviate usually set at 1.96 which corresponds to 95% confidence interval (value for error)
p= Taken as 50%=0.5 (According to the Centers for Disease Control, recent studies place hand hygiene adherence in hospitals between 29 percent and 50 percent. No known previous study was found in Bangladesh. As such I considered the sample proportion or percentage of incidence or prevalence as 50%.)
q=1-p (1-.5=.5)
d: Accepetable error ( Level of statistical significance) i.e. 0.05.
n = 384 (approx)
According to this formula my sample size need to be minimum 384, To reflect important variations in the populations and provide better level of accuracy I will take 384 samples as my study populations.
Sampling technique
HCWs of Combined Military Hospital Dhaka was selected for sample collection. Responednts were selected as non probability type of purposive or judgement sampling technique for data collection. Here respondents were chosen based on purpose of the study. Selection made by human choice rather than at random. It involved studying entire population of limited groups or subset of the population i.e. selected persons of different designations. As it may not represent the larger population, but it exactly needed to represent limited group of different types of subgroups of HCWs of CMH Dhaka. For proper representation and fulfill the purpose of the study almost half of the working population of different subgroups of HCWs of selected places / departments were taken as respondents.
Pre-testing
In order to determine the validity and reliability of the questionnaire, a pre-test study of the was done in CMH Rajendrapur.
Research approach
The purpose of the study was explained to the respondents prior to data collection and consent was taken accordingly. Prior to that the researcher oriented the healthcare workers of the Hospitals with clear instructions for replying the questionnaire correctly.
Data collection tools
A questionnaire was prepared as data collection tool containing the structured and semi-structured questions according to the objectives and variables of the study. Pre-testing was done at CMH Rajaendrapur. Modifications was done after pre-testing. The primary points considered in preparing the questionnaire form were as follows :
- Most of the questionnaire consists of close-ended questions. In brief, the main principle in preparing the questionnaire is to develop a questionnaire form starting from to make it suitable for all categories of HCWs educated to uneducated persons.
- Demographic, Socio-economic, knowledge, practice and available decontamination facilities information were included.
Data management and analysis
All interviewed questions was checked for its completeness, correctness and internal consistency to exclude missing or inconsistent data. The data was analyzed by using software SPSS version 17. For analyzing data some descriptive statistics was used. In order to find out association between dependent variable and a host of independent variables, chi square and Logistic regression tests were done. The data was also presented in tables and chart.
Data Presentation
Data was presented in the form of tables, graphs and charts in the result section of. this study.
Ethical consideration
Since data were collected from directly from the respondents, so there was direct relation or contact between respondents with researcher. Therefore there were questions of ethical implication; it was cautiously kept in mind about ethical matter which is related to researcher himself.
RESULTS
Study was descriptive type and conducted at CMH Dhaka from February 2011 to June 2011 and respondents were 384 heath care professionals.
Frequency distribution of different socio demographic characteristics of HCWs are as follows:
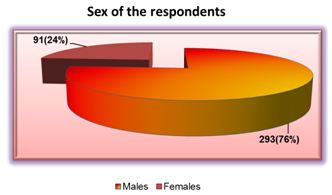
Figure 1: Pie diagram showing distribution of respondents as per sex (n=384)
This figure reveals that majority of the respondents 293(76%) are males and 91(24%) are females.
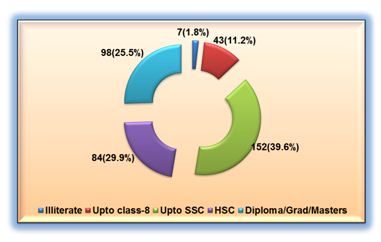
Figure 2: Diagram showing distribution of respondents educational status (n=384)
This figure reveals that educational level of the respondents were illiterate- 7(1.8%), upto class 8- 43(11.2%), upto SSC-152(39.6%), HSC-84(21.5%), Diploma / Graduate / Masters- 98(25.5%).
- Medical Assistant-97(25.3%), Operation theatre Assistant-62(16.1%), Nurses-73(19%), Dental Technicians-21(5.5%), Intensive Care Assistants-67(17.4%), Ward Boy-24(6.3%), Aya Cleaner- 29(7.6%), General Duty Assistant-11(2.9%).
- out of 384 respondents 259(67.7%) were married and 125(32.3%) were unmarried.
- out of 384 respondents 358(93,2%) were muslim and 26(6.8%) were hindu in religion.
- according to place of work respondents were from ICU-72(18.8%), Adult ward-70(18.2%), Paediatric Ward- 49(12.8%), OT-77(20.1%), OPD- 66(17.2%), Labour Ward- 50(13%).
| Table 2. Statistical analysis of selected socio demographic characteris of HCWs. | ||
Statistics of age and length of the service | ||
| Age in years | Length of service in years | |
| Mean | 32.40 | 11.51 |
| Median | 32.00 | 11.00 |
| Std. Deviation | 7.366 | 7.155 |
| Range | 38 | 29 |
| Minimum | 20 | 1 |
| Maximum | 58 | 30 |
Table 2 related to Mean age of respondents were 32.40 years, mean length of service was 11.51 years. Median age 32 years and median length of service 11 years. So age distribution were even – difference between mean and median is very narrow. SD of age was 7.366 and SD of length of service was 7.155. This SD was due to inclusions of some extreme age persons. Minimum age of respondents was 20 and maximum was 58. Minimum service length was 1 and maximum was 30 years.
This figure reveals that 104(27.1%) respondents are from age group 31-35 years, 84(21.9%) are both from age group below 25 years and 36-40 years, 69(18%) respondents are from 26-30 years age group and 43(11.2%) are of more than 40 years of age.
| Table 3. Distribution of respondents by compliance of hand decontamination and selected socio demographic factors | |||||||
Variables | Compliance of hand Decontamination | Total n= 384(100%) | df | P value
| |||
Yes(122) (31.8%) | No(262) (68.2%) | ||||||
| Sex | |||||||
Male106(27.6%)187(48.7%)293(76.3%)
1
.001
Female16(4.2%)75(19.5%)91(23.7%)Total122(31.8%)262(68.2%)384(100%)DesignationMedical assistant33(8.6%)64(16.7%)97(25.3%)
7
.002
Operation theatre assistant54(14.1%)8(2.1%)62(16.1%)Nurse16(4.2%)57(14.8%)73(19.0%)Dental technician17(4.4%)4(1%)21(5.5%)Intensive care assistant2(.5%)65(16.9%)67(17.4%)Ward boy0(0%)24(6.3%)24(6.3%)Aya/ cleaner0(0%)29(7.6%)29(7.6%)General duty assisstant0(0%)11(2.9%)11(2.9%)Total122(31.8%)262(68.2%)384(100%)EducationIlliterate0(0%)7(1.8%)7(1.8%)
4
.011
upto class 80(0%)43(11.2%)43(11.2%)upto SSC51(13.3%)101(26.3%)152(39.6%)HSC47(12.2%)37(9.6%)84(21.9%)Diploma/graduate/Masters24(6.3%)74(19.3%)98(25.5%)Total122(31.8%)262(68.2%)384(100%)Work PlaceICU0(0%)72(18.8%)72(18.8%)
5
.005
Adult ward22(5.7%)48(12.5%)70(18.2%)Paediatric ward15(3.9%)34(8.9%)49(12.8%)OT60(15.6%)17(4.4%)77(20.1%)OPD24(6.3%)42(10.9%)66(17.2%)Labor ward1(.3%)49(12.8%)50(13%)Total122(31.8%)262(68.2%)384(100%)
Table 3 related to distribution of respondents by compliance of hand decontamination and selected socio demographic factors.
There was significant association between sex of the respondents and compliance of hand decontamination.
The relationship between designation and compliance of hand decontamination was found to be significantly associated (p<,05).
It was evident also that from the result that all the respondents who are educated had compliance level on hand decontamination had significantly associated.
Significant association was found between work place and compliance on hand decontamination (p<.05).
| Table 4. Frequency distribution of different knowledge related factors of HCWs. | |||||
Knowledge about need of hand decontamination | |||||
Frequency | % | ||||
| Before and after patient care | 336 | 87.5 | |||
| Only after patient care | 30 | 7.8 | |||
| Only before patient care | 5 | 1.3 | |||
| Before going home after completion of duty | 13 | 3.4 | |||
| Total | 384 | 100.0 | |||
Knowledge about use of wash material( multiple answer given) | |||||
Frequency | % | ||||
| Soap &water | 374 | 97.4 | |||
| Alcohol mixed substance | 93 | 24.2 | |||
| Only by water | 9 | 2.3 | |||
| Other materials | 8 | 2.1 | |||
Knowledge about benefitted person | |||||
Frequency | % | ||||
| Good for patient | 21 | 5.5 | |||
| Good for HCW | 65 | 16.9 | |||
| Good for both | 297 | 77.3 | |||
| Don,t know | 1 | .3 | |||
| Total | 384 | 100.0 | |||
Knowledge about availability of facilities | |||||
Frequency | % | ||||
| Available | 383 | 99.7 | |||
| Not available | 1 | .3 | |||
Knowledge about source of infection ( multiple answers given) | |||||
Frequency | % | ||||
| Own uncleaned hand | 227 | 59.1 | |||
| Patient’s hand | 137 | 35.7 | |||
| From respiration | 170 | 44.3 | |||
| While talking | 4 | 1 | |||
| Other causes | 60 | 15.6 | |||
Source of knowledge | |||
Frequency | % | ||
| From teacher/ doctors | 297 | 77.3 | |
| From hospital authority | 64 | 16.7 | |
| From colleagues | 17 | 4.4 | |
| Others | 6 | 1.6 | |
| Total | 384 | 100.0 | |
Knowledge about requirement of time | |||
Frequency | % | ||
| 20 sec | 317 | 82.6 | |
| 10 sec | 33 | 8.6 | |
| dont know | 34 | 8.9 | |
| Total | 384 | 100.0 | |
Knowledge related factors
When enquired when to decontaminate hands, 336(87.5%) answered both before and after patient care, 30 (7.8%) replied only after patient care, 5 (1.3%) only before patient care, 13 ((3.4%) before going home after completion of duty.
When asked about the suitable material required to decontaminate respondents gave multiple answers, 374 (97.4%) replied both soap and water, out of them 93(24.2%) also included Alcohol mixed substance with soap and water as suitable material, 9(2.3%) told water was enough, and 8(2.1%) included other materials with soap and alcohol mixed substance, only one replied other materials alone.
When enquired about the benefitted persons from hand decontamination 21(5.5%) respondents replied hand decontamination is good for patients only, 65 (16.9%) opined it is good for HCWs, majority i.e.297( 77.3%) told it is good for both patients and HCWs. One respondent told that it is unknown to him.
Most of the respondents 383(99.7%) have knowledge about availability of facilities less one individual.
To find out the knowledge about the source of infection, many respondents gave multiple answers, 227(59.1%) respondents accepted own uncleaned hand as one of the source of infection. 137(35.7%) respondents mentioned patient’s hand as another source, from respiration of others 170(44.3%) respondents also opined as another source, only 4(1%) respondents told source of infection may be while talking with others and 60(15.6%) respondents also included some other causes as source of infection.
The respondents replied that they have learnt about hand decontamination and hospital infection from teachers/ doctors- 297(77.3%), from hospital authority 64(16.7%), from collegues 17(4.4%) and 6(1.6%) from other sources.
Majority of the respondents 317(82.6%) opined correctly that time required to decontaminate hand was 20 sec, 33(8.6%) told 10 second is enough and 34(8.9%) respondents told they donot have any knowledge about the time required to decontaminate hand.
| Table 5 . Distribution of respondents by compliance of hand decontamination and selected knowledge related factors | ||||||
Variables | Compliance of hand decontamination | Total n= 384(100%) | df | P value | ||
Yes(122) (31.8%) | No(262) (68.2%) | |||||
| Requirement of time | ||||||
20 Sec112(29.2%)205(53.4%)317(82.6%)
2
.001
10 Sec10(2.6%)23(6%)33(8.6%)Don’t Know034(8.9%)34(8.9%0Total122(31.8%)262(68.2%)384(100%)Knowledge about need of hand decontaminationBefore and after patient care118(30.7%)218(56.8%)336(87.5%)
3
.002
Only after patient care4(1%)26(6.8%)30(7.8%)Only before patient care05(1.3%)5(1.3%)Before going home after completion of duty013(3.4%)13(3.4%)Total122(31.8%)262(68.2%)384(100%)Knowledge about wash materials (multiple answer given)Soap &water122(31.8%)252(65.6%)374(97.4%)
1
.021
Alcohol mixed substance48(12.5%)45(11.7%)93(24.2%)
1
.001
Only by water09(2.3%)9(2.3%)
1
.031
Other materials1(.3%)7(1.8%)8(2.1%)
1
.219
Total122(31.8%)262(68.2%)384(100%)
| Knowledge about source of infection (multiple answer given) | Own uncleaned hand | 94(24.5%) | 133(34.6%) | 227(59.1%) | 1 | .001 |
| Patient’s hand | 55(14.3%) | 82(21.4%) | 137(35.7%) | 1 | .006 | |
| From respiration | 20(5.2%) | 150(39.1%) | 170(44.3%) | 1 | .002 | |
| While talking | 0 | 4(1%) | 4(1%) | 1 | .215 | |
| Other causes | 22(5.7%) | 38(9.9%) | 60(15.6%) | 1 | .229 | |
| Total | 122(31.8%) | 262(68.2%) | 384(100%) | |||
| Knowledge about benefitted persons | Good for patient | 5(1.3%) | 16(4.2%) | 21(5.5%) | 3 | .001 |
| Good for HCW | 6(1.6%) | 59(15.4%) | 65(16.9%) | |||
| Good for Both | 111(28.9%) | 186(48.4%) | 297(77.3%) | |||
| Don,t know | 0 | 1(.3%) | 1(.3%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) | |||
| Source of knowledge | From teacher/ doctors | 109(28.4%) | 188(49%) | 297(77.3%) | 3 | .038 |
| From hosp authority | 7(1.8%) | 57(14.8%) | 64(16.7%) | |||
| From colleagues | 0 | 17(4.4%) | 17(4.4%) | |||
| Others | 6(1.6%) | 0 | 6(1.6%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) |
Table 5 related to distribution of respondents by compliance of hand decontamination and selected Knowledge Related Factors are as follows:
P < 0.05. So association between knowledge related to requirement of of time for decontamination and compliance of hand decontamination is significant.
There was significant association between Knowledge about need of hand decontamination of the respondents and compliance of hand decontamination.
The relationship between Knowledge about wash materials soap and water, alcohol mixed substance, water and compliance of hand decontamination was found to be significantly associated (p<,05).
Significant association was found between Knowledge about source of infection- own uncleaned hand, patient’s hand, from respiration and compliance on hand decontamination (p<.05). But no significant association was found between Knowledge about source of infection of infection while talking, other causes and compliance on hand decontamination (p>.05)
It was evident also that from the result that all the respondents who are knowledge about benefitted persons had compliance level on hand decontamination are significantly associated.
There was significant association between source of knowledge of hand decontamination of the respondents and compliance of hand decontamination.
| Table 6. Frequency distribution of respondents by compliance of hand decontamination | ||||||
Compliance of hand decontamination | ||||||
Frequency | Percent | |||||
| Yes | 122 | 31.8 | ||||
| No | 262 | 68.2 | ||||
| Total | 384 | 100.0 | ||||
Number of times of decontamination of hand | ||||||
Frequency | Percent | |||||
| Not a single time | 8 | 2.1 | ||||
| 1-3 times | 98 | 25.5 | ||||
| 4-6 times | 243 | 63.3 | ||||
| More than 11 times | 35 | 9.1 | ||||
| Total | 384 | 100.0 | ||||
Availability of facilities of hand decontamination | ||||||
Frequency | Percent | |||||
| Yes | 375 | 97.7 | ||||
| No | 9 | 2.3 | ||||
| Total | 384 | 100.0 | ||||
Hand decontamination before taking meal | ||||||
| Frequency | % | ||||
| Yes | 381 | 99.2 | ||||
| No | 3 | .8 | ||||
| Total | 384 | 100.0 | ||||
Distance of hand decontamination facilities | ||||||
Frequency | Percent | |||||
| within 10 metres | 174 | 45.3 | ||||
| 11-20 meters | 171 | 44.5 | ||||
| More then 20 metres | 39 | 10.2 | ||||
| Total | 384 | 100.0 | ||||
Availability of decontamination materials( multiple answers given) | ||||||
| Frequency | % | |||||
| Whether Soap is available or not | 357 | 93 | ||||
| Whether Alcohol Mixed Substance is available or not | 139 | 36.2 | ||||
| Water is Available or not | 384 | 100 | ||||
Compliance related factors
Only 122 ( 31.8%) replied that they practice hand decontamination regularly, amongst them 293(76.3%) are male HCWs, practice amongst female HCW found very low, out of 16(4.2%) nurses 16(4.2%) nurse gave affirmative reply, out of 97(25.3%) Medical Assistants 33(8.6%) decontaminate regularly.
Compliance was higher among Operation Theatre Assistants 54(14.1%) out of 62(16.1%) and Dental Technicians 17(4.4%) out of 21(5.5%), those operation theatre asst works outside OT they are not practicing adequately.
Most alarming finding is all the Intensive Care Assistants believes that the use of gloves obviates the use of hand hygiene. Moreover they are using same gloves for several patients which is much hazardous, believing gloves are for own safety. They neither use gloves nor follow decontamination practices while usual work in ICU. Compliance was higher during procedures that carry a low risk for contamination and when the required intensity of care is greater. Hand washing compliance of health care workers in the Intensive Care Unit of CMH Dhaka is poor compared to other places of the hospital. Compliance was lower among nurses than other health care personnel, Patients in intensive care units are at gteater risk of nosocomial infection than those elsewhere in hospitals since they are already critically ill, are subject to multiple hand contacts from a wide variety of staff have indwelling invasive therapeutic and monitoring devices, and often receive broad-spectrum antimicrobial therapy.
Compliance of hand decontamination found extremely low amongst Ward boy, Aya/Cleaners and General Duty Assistants.
As per place of work it is found that in Adult ward 22(5.7%) out of 70(18.8%) HCWs, Paediatric ward 15(3.9%) out of 49(12.8%) HCWs, OT- 60(15.6%) out of 77(20.1%), OPD 24(6.3%) amongst 66(17.2%), labor ward 1(.3%) out of 50 (13%) decontaminate their hands properly- which is no doubt a poor performance.
About the cause of not practicing proper decontamination instructions respondents showed several remarkable reasons- 127(46%) respondents told because of time constraints they don’t wash hand, 106 (38.4%) persons told that they believes that use of gloves obviates the use of hand hygiene, 24(8.7%) showed reasons of facilities are inconveniently located, 10(3.6%) because of lack of facilities and 5(1.8%) for lack of knowledge. It was found that high demand for hand cleansing, which reflects high workload was associated with low compliance. Requirement for hand washing were much more frequent during busier times of the day and duing care of critically ill patients.
About number of times of decontamination during each shift of duty which comprises of 8 hours long, 243(63.3%) told they wash 4-6 times, 98 (25.5%) respondents wash 1-3 times, 35 (9.1%) more than 11 times and 8(2.1%) don’t decontaminate even a single times in a shift of duty.
Most of the respondents i.e. 387(99.2%) of all categories told that they wash their hands before taking meals. 383 (99.7%) told washing facilities are available in their duty places. 174(45.3%) respondents told facilities are within 10 metres, 171(44.5%) told facilities are 11-20 metres, 39(10.2%) told facilities are are more than 20 metres away from their duty places. 357(93%) respondents told that soaps are available, and 139(36.2%) respondents also confirmed availability of Alcohol mixed substances with soaps.
| Table 7. Distribution of respondents by compliance of hand decontamination and selected other compliance related factors | ||||||
Variables | Compliance of hand decontamination | Total n= 384(100%) | df | P value
| ||
Yes(122) (31.8%) | No(262) (68.2%) | |||||
| Cause of not following decontamination practices | Because of lack of knowledge | 4(1.4%) | 1(.4%) | 5(1.8%) | 5 | .002
|
| Because of time constraints | 6(2.2%) | 121(43.8%) | 127(46%) | |||
| Because of lack of facilities | 0 | 10(3.6%) | 10(3.6%) | |||
| Because of irritation or dryness of hands | 4(1.4%) | 0 | 4(1.4%) | |||
| facilities are inconveniently located | 0 | 24(8.7%) | 24(8.7%) | |||
| Beliefs that use of gloves obviates the use of hand hygiene | 0 | 106(38.4%) | 106(38.4%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) | |||
| Number of times practices hands decontamination | Not a single time | 1(.3%) | 7(1.8%) | 8(2.1%) | 3 | .191 |
| 1-3 times | 26(6.8%) | 72(18.8%) | 98(25.5%) | |||
| 4-6 times | 80(20.8%) | 163(42.4%) | 243(63.3%) | |||
| More than 11 times | 15(3.9%) | 20(5.2%) | 35(9.1%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) | |||
| Hand wash practice before taking meal | Yes | 122(31.8%) | 259(67.4%) | 381(99.2%) | 1 | .316
|
| No | 0 | 3(.8%) | 3(.8%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) | |||
| Availability of facilities | Yes | 122(31.8%) | 261(68%) | 383(99.7%) | 1 | .682 |
| No | 0 | 1(.3%) | 9(.3%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) | |||
| Distance from facilities | within 10 metres | 70(18.2%) | 104(27.1%) | 174(45.3%) | 2 | .001 |
| 11-20 meters | 52(13.5%) | 119(31%) | 171(44.5%) | |||
| More then 20 metres | 0 | 39(10.2%) | 39(10.2%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) | |||
Table 7 related to distribution of respondents by distribution of respondents by compliance of hand decontamination and selected other compliance related factors are as follows:
P <0.05.So association between cause of not following decontamination and compliance of hand decontamination is significant.
There was no significant association between number of times practices hands decontamination and compliance of hand decontamination.
There was no significant association between hand wash practice before taking meal and compliance of hand decontamination.
The relationship between availability of facilities and compliance of hand decontamination was found to be not significantly associated (p>,05).
Significant association was found distance from facilities and compliance on hand decontamination (p<.05).
| Table 8. Distribution of respondents by compliance of hand decontamination and availability of selected decontamination materials. | ||||||
Variables | Compliance of hand decontamination | Total n= 390(100%) | df | P value
| ||
Yes (123) (31.8%) | No (267) (68.2%) | |||||
| Availability of soap | Yes | 121(31.5%) | 236(61.5%) | 357(93%) | 1 | .001
|
| No | 1(.3%) | 26(6.8%) | 27(7%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) |
| ||
| Availability of alcohol mixed subsatnce | Yes | 58(15.1%) | 81(21.1%) | 139(36.2%) | 1 | .001 |
| No | 64(16.7%) | 181(47.1%) | 245(63.8%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) |
| ||
| Availability of water | Yes | 122(31.8%) | 262(68.2%) | 384(100%) | – | –
|
| No | 0 | 0 | 0 | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) |
| ||
| Availability of facilities | Yes | 122(31.8%) | 261(68%) | 383(99.7%) | 1 | .682 |
| No | 0 | 1(.3%) | 1(.3%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) | |||
| Distance of available facilities | within 10 metres | 70(18.2%) | 104(27.1%) | 174(45.3%) | 2 | .001 |
| 11-20 meters | 52(13.5%) | 119(31%) | 171(44.5%) | |||
| More then 20 metres | 0 | 39(10.2%) | 39(10.2%) | |||
| Total | 122(31.8%) | 262(68.2%) | 384(100%) | |||
Table 8 related to distribution of respondents by compliance of hand decontamination and availability of selected decontamination materials are as follows:
P <0.05.So association between availability of soap and compliance of hand decontamination is significant.
There was no significant association between availability of alcohol mixed subsatnce and compliance of hand decontamination.
There was no significant association between distance of available facilities compliance of hand decontamination.
Focus Group Discussions
The Focus Group Discussion took place at CMH Dhaka in observation and interviewing methods, contributing to subsequent modifications of the interview guide/questionnaire used.
A total of 10 health workers including nurses, Medical assistants working in CMH Dhaka; Participants joined the study on a voluntary basis. Information about the purpose of the study was given to the hospital health workers, and verbal consent was obtained from those who participated. No incentives and/or rewards in cash or in kind were offered to the study participants.
Focus group discussion facilitators, fixed a set of three questions (listed below) to guide their group discussions. Focus group discussion was conducted in Conference room of Gynae Ward. Nurses and Medical assistant formed focus group discussion group, they were mixed in terms of gender had majority women.
Semi-structured interviews were conducted individually with the 10 health workers who participated in focus group discussions, in the obstetric wards. The study design and methods provided a robust system of data quality check through triangulation of information by sources and methods.
Study participants and methods
Six Nurses (all women) and four medical assistants ( All men).
Discussion
All focus group discussion participants (n=10). were discussed the questions amongst themselves.
Observations
A total of 30 observations were documented
Facilitator/moderator’s guide/questionnaire :
- Why is hand decontamination practice so poor?
- What makes the performance poor?
- What can be done to improve the situation?
Focus Group Discussion findings:
Reasons for inadequate hand decontamination practices
Nurses
- Not enough sinks
- Inconveniently located sinks
- Insufficient time
- No towels/ paper towels
- Lack of hand lotions /lubricants
- Lack of water
- Poor quality of soap/ common soap used by all
- Lack of funds
- They did not wash their hands was because they did not like to use common towels to dry their hands after washing.
Medical Assistants
- Not enough sinks
- Inconveniently located sinks
- Not enough soap and hand lotions /lubricants
- High workload and understaffing
- Insufficient time
- Ignorance of guidelines – Lack of knowledge of guidelines and protocols
- Negative influence of colleagues on hand hygiene compliance
- Perceived lack of scientific information demonstrating the impact of improved hand hygiene on hospital infection rates
- Skin irritation or sensitivity
- Interference with worker–patient relationship
- Patient needs take priority
- No role model from colleagues or superiors
- Scepticism about the value of hand hygiene
What makes the performance poor?
They prefer to wear gloves, and admitted that they do not change their gloves between patient contacts. Those who preferred gloves saw them as protective devices for themselves, rather than for their patients. These same health workers considered hand decontamination to be time consuming.
Behaviour issues
- Forgetfulness
- Low priority by some HCWs
- Wearing gloves — perception that this negates the need for hand hygiene
- Lack of motivation — lack of educational resources, guidelines and protocols
- Translation of behaviour learned and practised in the community
- Not monitored or supervised by doctors
Observed risk factors for poor adherence
- Working in intensive care
- Wearing gowns/gloves
- Activities with high risk of cross-transmission
- Understaffing or overcrowding
- High number of opportunities for hand hygiene per hour of patient care
- Nursing assistant status
OUTPUT
- Perceived lack of time
- Lack of motivation
- Negligence
What can be done to improve the situation?/ What steps have been taken to improve performance?
- Regular and sufficient supplies of protective materials including cleaning agents, liquid soap.
- Facilitative supervision and follow-up are necessary
- Campaign on Keeping Hands Safe
- Ingredients of success in implementing prevention of Hospital Acquired Infection.
- Motivation and development of consciousness amongst patients and attendants..
- Staff believed their hand hygiene behaviour was easier to maintain. Typical comments included the following:
- It becomes routine, habit.
- It is ingrained behaviour.
- It’s easier to maintain hand rub than hand-washing.
Proposed Campaign CLEAN HANDS WILL SAVE strategies
- Provision of alcohol-based hand rub at point of patient care
- Giving incentives to staff and leaders
- Involvement of patients, attendants and visitors in the hand hygiene aspect of their care
- Promotion of collateral material to market the campaign and maintain the interest of target groups in the campaign messages:
- Talking walls — staff posters .
- Hand hygiene technique poster
- Patient/visitor targeted posters
- Patient/visitor brochures
- Stickers
- T-shirts with campaign logo
- Balloons with campaign logo
Tips for hospital patients suggested by the participants
- “Patients Best Defense” “Since human error may account for millions of infections contracted in hospitals each year , patients should take an active role in protecting themselves. A few tips to help ensure that they leave the hospital at least as healthy as they have arrived:
- When visiting doctors, don’t be embarrassed to ask whether former patients have contracted infections.
- Hand washing can kill up to 99 per cent of all known bacteria. Insist that HCWs wash their hands before touching patients.
- Patients accompanied by family or friends who keep an eye on their care are less likely to fall prey to mishaps.
- Catheters and IVs are common point of entry for germs; they should be removed when no longer needed. Ask whether it’s time to take them out.
- Overuse of antibiotics is one of the most dangerous factors behind hard-to-treat infections. Ask whether antibiotics are right for the patients and take them exactly as prescribed.”
Conclusion
An important strength of this Focus Group Discussion is its participatory design which has served to engage hospital-based health workers from all levels of service in problem solving activities and processes. This study was iterative and dynamic, rather than linear and static. Thus, to formulate policy for CMH representing stakeholders who are inclined to take it upon themselves to raise the standards of quality of health care as they develop a stronger knowledge base of infection prevention and the necessary skills to implement agreed procedures, is a logical outcome of the study. The study design and methods are robust and replicable, paving the way for more such studies to be conducted in order to build a strong, reliable and useful evidence-base for policy and practice concerning nosocomial infections that may be applicable to other Armed Forces Hospitals.
Discussion
Hand decontamination is a simple and effective means of preventing the spread of infection abound. For example, few years back in Bangladesh National Public/ private media announcements included reminders to wash their hands thoroughly in order to prevent the spread of seasonal infections, particularly influenza. Similarly, in the Far East, public health advice concerning the outbreaks of viral infections such as SARS have pointed the lay person in the direction of the simple practice of handwashing with soap and water, especially in children’s day care centers, pre-schools, schools, shopping malls and other public places. Hospital nurses, doctors and consultants are rarely mentioned in public as likely vehicles of the spread of infection in health care settings; and yet they are, if they fail to wash their hands routinely and thoroughly. In many developed countries, patients are advised to demand that their doctors wash their hands before they touch them. However, such advice is not very practical for those in ICUs hitched to ventilators, intra-venous drips and catheters; or those who are under anesthetics undergoing surgery or otherwise unconscious. Dissemination of infection prevention information and advice concerning hospital acquired infection is probably best carried out in the community, away from hospitals, so that lay people can prepare themselves for hospitalization well in advance. To this effect, a renowned University Professor (Elaine Larson) was pictured in her laboratory with her microscope and test tubes in a fairly recent issue of the glossy magazine PEOPLE, with a caption that read “Nobody washes their hands enough”. This included five tips under “Patients’ Best Defense”. So, can hospital patients in western countries take legal recourse for hospital acquired infection? Anecdotal evidence suggests that at least some relatives of deceased patients do, but may not succeed in establishing the cause of death beyond reasonable doubt.
In this study only verbal answer for complince was recorded. No observational data was taken. Self reporting overestimates compliance. After unobtrusive observation of HCW to obtain a baseline hand washing rate, like in a study by Tibballs asked a sample to estimate their own hand washing rates before patient contact. Their perceived rate of 73% (range 50%95%) contrasted sharply with the observed frequency of just 9%. Pritchard and Raper were astonished that “doctors can be so extraordinarily selfdelusional about their behaviour.”
In a study by Pittet D, Mourouga P, Perneger TV, Members of the Infection Control Program. Compliance with handwashing in a teaching hospital. Ann Intern Med 1999;130:126–302,834 observed opportunities for hand hygiene, average compliance was 48%. In another observational studies of hand-hygiene adherence- Adherence of HCWs to recommended hand-hygiene procedures has been poor, with mean baseline rates of 5%–81% (overall average: 40%) 95,97. In this study it was found 31.8% compliance of hand decontamination.
In the same study it was found that non compliance level is lowest amongst nurses, In another observational studies conducted in hospitals, HCWs washed their hands an average of five times per shift to as many as 30 times per shift 2946,93; certain nurses washed their hands <100 times per shift 90. In our study is also alarmingly low amongst nuses (16%)but higher than ward boy, aya and cleaners which is also obvious. Frequency of decontamination In that study nonadherence was higher in intensive-care units compared with internal medicine wards (OR: 2.0; 95% CI = 1.3–3.1), The higher the demand for hand hygiene, the lower the adherence; on average, adherence decreased by 5% (+ 2%) for each increase of 10 opportunities per hour when the intensity of patient care exceeded 10 opportunities per hour . Similarly, the lowest adherence rate (36%) was found in intensive-care units, where indications for hand hygiene were typically more frequent (on average, 20 opportunities per patient-hour)..- in our study compliance found much lower amongst intensive care assistants because of wrong conception about use of gloves which caused deviation in the result.
The highest adherence rate (59%) was observed in pediatrics wards, where the average intensity of patient care was lower than in other hospital areas (an average of eight opportunities per patient-hour). 18,19,106. In our case highest compliance rate was found in OT(20.1%) and then OPD(17.2%), labor ward (13%), paediatric ward(12.8%).
The findings of this study make two major contributions. Firstly, the study confirms that both cognitive and technical input are absent for proper hand decontamination – it is not an ‘either-or’ proposition. Promotion of hand decontamination is also relevant to OPD which do not accommodate in-patients. A study conducted in Belgium has shown that HCWs hand washing practice left more to be desired, highlighting the need to target them as well. However, the point of international comparison is not to encourage complacency among our doctors. The view that “even rich countries have this problem, so why bother?” needs to be tackled head on. In CMH Dhaka, the challenge is to at least maintain previously documented and celebrated standards of cleanliness and patient care; if not to excel. Can HCWs who can’t be bothered to wash their hands get away with using gloves? In the case of CMH, HCWs preference to use gloves, and their failure to change them between patient contact seemed to be indicative of two basic misconceptions: one, that gloves are meant to protect the HCW, and not the patient; and two, that repeated use of the same pair of gloves does not matter to either the HCW, the patient, or for both. Studies have shown the use of gloves to be hazardous for both patient and health worker unless certain precautions are taken. Latex gloves can easily get punctured and/or provide ambient medium (moisture and warmth) for bacteria to breed rapidly. Washing hands and donning them with antimicrobial substances before gloving (particularly for unsterile vinyl gloves) and washing hands immediately after removing the gloves (even when using sterile gloves) is recommended, but much depends on the particulars of the hospital unit concerned. Unmentioned by the authors, this may be more a reflection of nurses’ perfection of the art of hand washing than the relative merits of sterile or non-sterile gloves versus hand washing. While the majority (72%) of CMH health workers were observed washing their hands for 10-15 seconds, only 31.5% washed their hands thoroughly. The technique used is clearly more important than the amount of time spent washing hands. Secondly, in Focus Group Discussion’s major contribution in the area of patient participation is important. A recent systematic review has highlighted the significance of patient involvement in quality of health care and service planning and improvement in the UK. The focus group discussion concluded that “the effects of involving patients were likely to be complex, affecting different aspects of services in different ways. The views of patients are among many factors that influence change in health services, and providers of health care remain the final arbiter of how much weight is attached to patients’ views.” According to Gill Walt’s careful analysis, so-called “Egalitarian-authoritarian” systems such as those of Cuba and China have been characterized by resource redistributive policy and practice, often with positive health effects such as declines in infant mortality. However, the findings of focus group discussion concerning patients’ views deserve careful consideration. Improvements in hand washing practice and general cleanliness of beds and disinfecting of the equipment and surroundings of patients’ beds requires patients participation. Patients’ voices definitely carry more weight in this regard as health workers may be expected to over-report good practice because of basic nature of members of Armed Forces personnels. Health workers’ training in infection prevention needs to incorporate their own protection as well. With particular reference to autopsy units, the use of gloves, even double layers and strong gauntlets for the nondominant hand are recommended. Health workers may also protect themselves with gloves from the hazards of ingesting deadly pathogens while reading and licking their fingers in order to turn the pages of hospital patient records. However, the most important lesson seems to be, HCWs: avoid treating gloves as protective devices against your patients. The costs of hospital acquired infection transmitted by contaminated gloves and hands are enormous for the patient – literally a matter of life and death – and they are also costly to the hospital, the Armed Forces and to the country as a whole as increased morbidity and mortality means loss of human, social, and economic capital. One study has documented the cost-effectiveness of simple preventive strategies such as health workers’ proper washing and scrubbing of hands to prevent and control nosocomial infections. This is relevant to Bangladesh and to other countries in similar stages. Hand decontamination between care of patients remains one of the most important measures for preventing the spread of pathogens from the hands of health care workers in hospitals.
My study confirms that the primary problem with hand washing is laxity of practice. HCWs freely acknowledge that hand washing is important in the control of nosocomial infection and suggest pressure at work, a lack of patient contact, detrimental effects of hand washing agents on skin and the more important needs of the patient as reasons for not practicing hand washing. during routine patient care, health care workers disinfected or washed their hands in approximately quarter of the indicated instances, I observed poor compliance rate among nurses compared with other health care workers, which is very unfortunate because nurses had the most opportunities to wash their hands. A disturbing finding was that activities that carried higher risk for transmission were associated with low compliance. In particular, compliance was very low for care between a dirty and a clean body site and while using gloves.
Conclusion
A modest measure—with big effects Hospital acquired infection damages patients, prolongs hospital stays, consumes scarce hospital resources, and thus presents a major challenge for clinical governance. In a seminal intervention study 150 years ago Semmelweis insisted that doctors performing necropsies washed their hands before delivering babies, so reducing mortality due to streptococcal puerperal sepsis from 22% to 3%.2 Many studies since have confirmed that HCWs decontaminating their hands between seeing patients can reduce hospital infection rates. Nevertheless, health care workers still fail to decontaminate their hands and fail to appreciate the importance of doing so. In this study show low compliance of hand decontamination in intensive care units, and as per designation /trade among Nurses and Intensive care Assistants. Though nurses spend more time than other HCWs in direct patient contact and may think that they need to decontaminate their hands less often, they have many transient contacts and move from ward to ward.
Why is compliance so poor? Even when taught the theoretical basis of hand washing, healthcare workers do not seem to understand the risks associated with noncompliance. Hospital acquired infections usually present as sporadic cases, perceived as insignificant or unrelated to noncompliance. Staff horrified by lice on a patient fail to consider the potentially far more serious consequences of bacteria present on their hands.
The failure of healthcare workers to decontaminate their hands reflects fundamentals of attitudes, beliefs, and behaviour, and there are no simple solutions. Attempts should be made to improve hand decontamination compliance through education, and indeed elementary hygiene practice should be taught explicitly in medical/para medical institutes. Principles taught in the lecture theatre can be reinforced by experiential learning, such as demonstrating the need for proper hand washing technique by showing microbial growth from unwashed hands and by using fluorescent oilbased dyes to illustrate the effectiveness of hand washing.
Such methods increase personal impact, but, though they may be temporarily improve compliance, behavioural changes tend not to be maintained Prevention and control of HAI remain major priorities for us and other countries around the world. It can effectively be advanced through the simple and inexpensive decontamination practice of proper handwashing between patient contact by doctors, nurses, health assistants and other support staff. Whatever their reason for failing to wash their hands at critical times, health workers who spread HAI stand guilty of violating the Hippocratic Oath, Do No Harm, that is sacred to their profession, and should therefore be made accountable in the most appropriate way. Rewarding good practice may be a useful step forward, however, ultimately, only the doctors (and nurses and other health workers) individual as well as collective motivation, sense of professional integrity, and pride in their own work can galvanize collective efficacy and accountability.
Greater evidence on the importance of hand decontamination to public health will also help improve acceptance. In particular, rigorous trials of the impact of hand decontamination on infectious disease are needed. The evidence for the impact on acute respiratory infections, specifically, is still weak and needs more investigation. ln addition more research on comparing the effectiveness of different approaches to generating behavior change will help optimize implementation.
Available evidence is sufficient to enable public health to act on issues that, on balances of probability, look the most promising. From this perspective, making Hand decontamination with soap ubiquitous is a key challenge for public health in the 21st century.
Government, industry, support agencies, and academia all have important roles to play. We found out that the average level of compliance with recommended hand washing practices among health care workers was 31.8%, which is low the range of compliance noted in previous studies conducted in different Hospitals of developed countries. Compliance was lower among nurses and Intensive Care Assistants other than health care personnel, findings not confirming previous observations. Several new findings of icular concern include the fact that HCWs beliefs that use of gloves obviates the use of hand hygiene and another is time constraints- ultimately understaffing. Despite the strict rules at CMH compliance is still very poor and re- evaluation with stricter implementation of its programs are needed. Reducing the time required for hand washing may make it feasible for caregivers with high workloads to decontaminate their hands more frequently and placement of alcoholic rinse or gel or similar products near each patient’s bed and at other locations that are eas’y accessible should be evaluated to determine whether this strategy results in improved hand hygiene’ controled studies are also needed to determine whether frequent use of alcohot-based hand rinses and gets by personnel will lead to improved control of nosocomial infection hospital administrators should strive to create an organizational atmosphere in which adherence to recommended hand hygiene practices is considered an integral part of providing service. There is a need to re-evaluate programs regarding strict hand washing in the CMH Dhaka.
Recommendations
Health-care worker educational and motivational programs:
- As part of an overall program to improve hand-hygiene practices of HCWs, educate personnel regarding the types of patient-care activities that can result in hand contamination and the advantages and disadvantages of various methods used to clean their hands.
- Monitor HCWs’ adherence with recommended hand-hygiene practices and provide personnel with information regarding their performance.
- To minitor CCTV cameras can also be used specially in important places like ICU, OT.
- Encourage patients and their families to remind HCWs to decontaminate their hands.
Administrative measures:
- Make improved hand-hygiene adherence an institutional priority and provide appropriate administrative support and financial resources.
- Implement a multidisciplinary program designed to improve adherence of health personnel to recommended hand-hygiene practices.
- As part of a multidisciplinary program to improve hand-hygiene adherence, provide HCWs with a readily accessible alcohol-based hand-rub product.
- To improve hand-hygiene adherence among personnel who work in areas in which high workloads and high intensity of patient care are anticipated, make an alcohol-based hand rub available at the entrance to the patient’s room or at the bedside, in other convenient locations, and in individual pocket-sized containers to be carried by HCWs.
- Store supplies of alcohol-based hand rubs in cabinets or areas approved for flammable materials.
Performance Indicators : The following performance indicators are recommended for measuring improvements in HCWs’ hand-hygiene adherence:
- Periodically monitor and record adherence as the number of hand-hygiene episodes performed by personnel/number of hand-hygiene opportunities, by ward or by service. Provide feedback to personnel regarding their performance.
- Monitor the volume of alcohol-based hand rub (or detergent used for handwashing or hand antisepsis) used per 1,000 patient-days.
- Monitor adherence to policies dealing with wearing of artificial nails.
- When outbreaks of infection occur, assess the adequacy of health-care worker hand hygiene.
Informed Decision Making Consent Form
- Informed Decision Making Consent: The research work done with providing the respondent the clear information about the subject & after they agreed willingly data/ information was taken.
- Information sheet: to provide the respondents the clear information about the research work, its aim & to interview the respondent. All the information kept private.
- No remuneration given to the respondent for participation and he / she could withdraw from the participation at -any time without any bindings, lf he/she agreed then the consent taken.
- Consent: The consent may be written or verbal depending on the educational background of the respondent. The written consent was as follows.
First Part of the post:
Thesis Paper on Compliance of Hand Decontamination of Combined Military Hospital Dhaka (Part-1)
Thesis Paper on Compliance of Hand Decontamination of Combined Military Hospital Dhaka (Part-2)