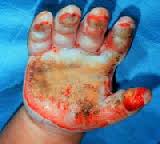
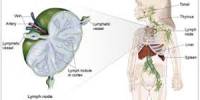
Burn causes coagulative necrosis of the epidermis and underlying tissues, with the depth depending on the temperature to which the skin is exposed and the duration of exposure. The specific heat of the causative agent also affects the depth.
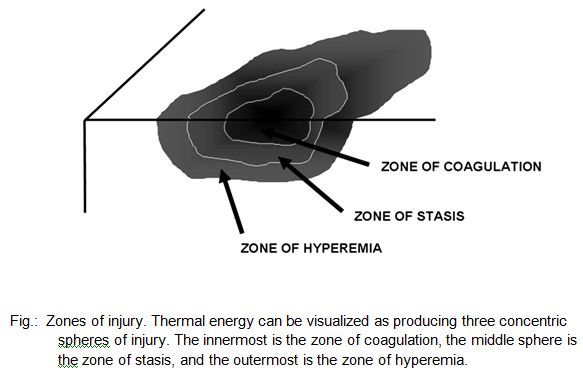
The microscopic pathologic feature of the burn wound is principally surface coagulation necrosis. Jackson’s 1947 state that burned tissue has three distinct zones1.12. The first is the zone of “coagulation,” or necrosis with irreversible cell death and no capillary blood flow. Surrounding this is a zone of injury or stasis, characterized by sluggish capillary blood flow and injured cells. Although damaged, the tissue is still viable. Further tissue
injury can be caused by products of inflammation such as oxidants and vasconstrictor mediators. Environmental insults such as hypoperfusion, desiccation, or infection can also cause the injured tissue to become necrotic. This process is called wound conversion. The third zone is that of “hyperemia,” which is the usual inflammatory response of healthy tissue to nonlethal injury. Vasodilatation and increased capillary permeability is typically present 8.12.
A rapid loss of intravascular fluid and protein occurs through the heat-injured capillaries. The volume loss is greatest in the first 6–8 hours, with capillary integrity returning toward normal by 36–48 hours. A systemic inflammatory response occurs in response to a large body burn, resulting in the release of oxidants and other inflammatory mediators into unburned tissues. This leads to a shift of extracellular sodium and water into the intracellular space. Smoke inhalation markedly increases the hemodynamic instability, fluid requirements, and mortality rates by adding another source of intense inflammation leading to local lung and systemic tissue damage.
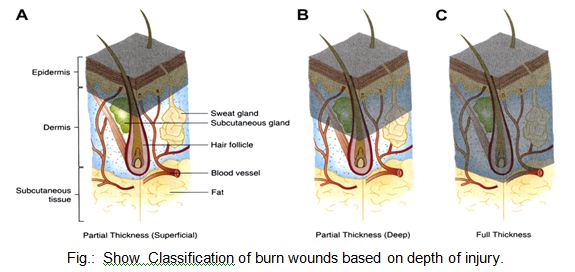
Traditionally in Thermal Injuries, burns have been classified as first-, second-, and third-degree, but the current emphasis on burn healing has led to classification as partial-thickness burns, which can heal spontaneously, and full-thickness burns, which require skin grafting, although deep partial-thickness burns are usually excised and grafted as well 2.8.12.
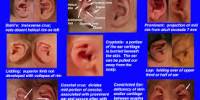
A first-degree burn involves only the epidermis and is characterized by erythema and minor microscopic changes; tissue damage is minimal, protective functions of the skin are intact, skin edema is minimal, and systemic effects are rare. Pain, the chief symptom, usually resolves in 48–72 hours, and healing takes place uneventfully. In 5–10 days, the damaged epithelium peels off in small scales, leaving no residual scarring. The most common causes of first-degree burns are overexposure to sunlight and brief scalding.
Second-degree or partial-thickness burns are deeper, involving all of the epidermis and some of the corium or dermis.
The systemic severity of the burn and the quality of subsequent healing are directly related to the amount of undamaged dermis. Superficial burns are often characterized by blister formation, while deeper partial-thickness burns have a reddish appearance or a layer of whitish, nonviable dermis firmly adherent to the remaining viable tissue. Complications from superficial second-degree burns are mainly severe pain related. These burns usually heal with minimal scarring in 10–14 days unless they become infected. Deep dermal burns heal over a period of 4–8 weeks .Conversion to a full-thickness burn by bacteria is common. Skin grafting of deep dermal burns, when feasible, improves the biologic quality and appearance of the skin cover.
Full-thickness (third-degree) burns have a characteristic white, dry, waxy appearance and may appear to the untrained eye as unburned skin. Burns caused by prolonged exposure to heat, with involvement of fat and underlying tissue, may be brown, dark red, or black. The Thermal Injuries diagnostic findings of full-thickness burns are lack of sensation in the burned skin, lack of capillary refill, and a leathery texture that is unlike normal skin. All dermal epithelial elements are destroyed, leaving no potential for reepithelialization.