Science, a biotechnology startup and rival of Neuralink, unveiled a new platform on Monday (march 13, 2023) with the goal of facilitating the speedy development and production of medical devices by other businesses.
The platform, known as Science Foundry, gives businesses access to more than 80 of the company’s products and services, including its thin-film electrode technologies, enabling them to make use of and expand upon Science’s internal infrastructure.
The cost of the technology required to develop medical devices is often “prohibitive” for early-stage startups, Science Co-Founder and CEO Max Hodak told CNBC in an interview. Individual tools can cost anywhere from $200,000 to $2 million, and Hodak said companies could easily spend hundreds of millions building a manufacturing line.
For many startups, that cost is too much to bear, but Hodak is hoping Science Foundry can help.
“Hopefully, we bring down the barriers to innovation,” Hodak said. “There’s a bunch of smart people out there that have a bunch of different ideas than the ones that we have, and we would like to enable them.”
Science is part of the growing brain-computer interface, or BCI, industry. A BCI is a system that deciphers brain signals and translates them into commands for external technologies. Perhaps the best-known name in the space is Neuralink, thanks to the high profile of founder Elon Musk, who is also the CEO of Tesla, SpaceX and Twitter.
Hodak co-founded Neuralink and served as the company’s president until he announced his departure in 2021. Hodak worked on the creation of a BCI system at Neuralink that is intended to be implanted directly into the brain, but he is currently developing an implant at Science that doesn’t even touch the brain.
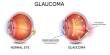
The Science Eye, a visual prosthesis designed to help people with two serious forms of blindness return some visual input to their brains, is Science’s premier BCI system.
The Science Eye uses a micro-LED array that is surgically implanted over the retina and is extremely thin and flexible. The optogenetic gene therapy used by Science to change a set of light-sensitive cells in the optic nerve is controlled by the implant. When one pixel in the array turns on, a cell in the optic nerve turns on as well, which can be used to drive the nerve and transmit visual information to the brain.
Special glasses that are outfitted with tiny sensors and cameras power Science’s implant. The LED array translates the images it receives from the glasses and sends them up to the optic nerve.
Hodak said the resulting images will look different than what people with healthy eyes are used to at least for the first iteration of the technology but that it will be very restorative for patients with no light sensitivity. Eventually, he said thinks Science will be able to reproduce high-resolution color vision.
Science has been testing the technology in rabbits, and Hodak said the company hopes to eventually conduct trials with human patients as soon as next year.
The business’s next platform, Science Foundry, is designed to assist businesses with similarly ambitious concepts. Hodak said he expects to see demand from other neurotechnology companies, but that other medical technology startups and even quantum computing companies represent growth opportunities.
The cost of using Science Foundry is comparable to the cost of working with academic facilities, which are “cheap to get started,” Hodak said. But while academic facilities typically do not allow companies to test devices on patients or sell them on the market, Hodak said it will be easier for Science Foundry customers to commercialize their products.
Hodak said the platform will benefit Science and the broader industry as a whole.
“This enables us to afford larger-scale and more capabilities that then we can use to enable the community and ourselves even further,” he said.