Discussions of the results:
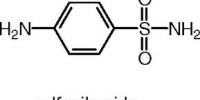
Quality and quantity are two most important parameters for any pharmaceutical dosage forms. So, quality control is not only a vital task for the manufacturers but also an important global concern. This project work was an approach to estimate the quality of marketed capsule preparations of Tetracycline HCl which is most commonly known as antibiotic to the consumers.
The proposed method was successfully applied for the qualitative analysis of Tetracycline HCl by determining the potency of capsules dosage forms of different brands.
As tetracycline is the most common antibiotic drug that fights bacteria in the body taken by people; it must have to be safe & effective. Several brand of sample was collected from the market in capsule dosage forms.
The experiment was done by trial and error method. Ten brands of Tetracycline HCl capsules manufactured by local pharmaceutical companies were collected from the local market for potency determination. Collected capsules were coded as TET001, TET002, TET003, TET004, TET005, TET006, TET007, TET008, TET009 and TET010.
From the results and observation of the project work some conclusions have been drawn. As the numbers of samples were small, the result may somewhere be misleading or incomplete. Again, there was always some variation in the results which may occur due to various factors like environmental factors, experimental errors, personnel error etc.
Both physical and chemical parameters were tested. The physical parameters were complied with the specifications; though a few brands deviated a little.
On the contrary, the test results of capsules showed effective in respect of raw materials; but the test results of some brands deviated from the values of raw materials. Though several samples were studied but only reproducible results obtained from capsules have been mentioned in this project report.
Quality of tetracycline hydrochloride raw material is the most important factor affecting potency of the products. Normally, general appearance is not a crucial parameter to check the performance of capsule formulation. The performance of any dosage form especially capsule depends largely upon various physical and chemical parameters.
Weight variation test were performed of each brand of capsule and they were found uniform in weight. Only two brands from them were deviated from the standard weight.
The disintegration time of the capsule of each brand was within the British Pharmacopoeia (BP) specification.
Beside the marketed samples (capsules) test was also performed for the pure sample (pure tetracycline hydrochloride), and the results were found to meet the official specification of British pharmacopoeia (BP). The standard value obtained from first sample was met for the comparison of the results obtained from the dosage form.
All the tests were performed very carefully by validated method, calculated, and recorded.
Conclusion
Bangladesh is a developing country. Most of the area of Bangladesh there is inadequate facility for proper quality control of the health related pharmaceutical products. Proper quality control is one of the prime precautions of health related products. Proper quality control according to validated official method is expensive, time consuming and difficult to practice. Quality control according to official method needs trained and skilled personals and well equipped laboratories which are not amenable in Bangladesh.
Furthermore, the laboratory facilities for assaying drugs are limited especially in remote areas. For these reasons, spectrophotometric method is recommended. Though it is not a time consuming process but it is expensive but not difficult to operate.
Though it cannot replace any official method, it can still be used as a very effective method of secondary screening of drugs.
In-vitro evaluation of tetracycline hydrochloride is a very simple and rapid means of selecting best formulation among available brands in the market. For better product development, tetracycline hydrochloride manufacturer should consider the test for potency of the raw materials used while formulating capsules.
In this analytical work some results of the samples were not reproducible, hence could not be shown.
From the above discussion it can be concluded that the available marketed drugs in Bangladesh should be screened properly and successfully by using this portable spectrophotometric method. Hospital pharmacist should also carry out this test before selecting an ideal product for hospital use.