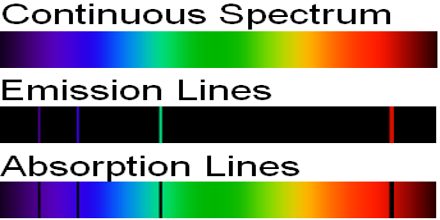
Kirchhoff’s Laws said: Light of all wavelengths shines on an atom; only light of an energy equal to the difference between “floors” will be absorbed and cause electrons to jump up in floors. The rest of the light passes on by to our detector. We see an absorption spectrum: light at all wavelengths minus those specific wavelengths. This lecture briefly describe Kirchhoff’s Laws of Spectral Lines. Spectral lines are a dark or bright line in an otherwise uniform and continuous spectrum, resulting from emission or absorption of light in a narrow frequency range, compared with the nearby frequencies.
Kirchhoff’s Laws of Spectral Lines